Reproduction
The chapter about the reproduction is a compilation of three articles, two of them originally included in: Kempkes, M. et al., (2013): Beiträge zur Biologie und zum Artenschutz der Hochlandkärpflinge, Neue Brehm-Bücherei, Band 681, Westarp Wissenschaften. ISBN: 9783894322625. The first article is a (shortened) translation of the 10th chapter of this book with the original German title "Zur Fortpflanzungsbiologie von Hochlandkärpflingen" (engl.: On the reproductive biology of Goodeids) from Hartmut Greven, Institute of Cell Biology and Morphology of the Heinrich-Heine-University, Düsseldorf, Germany. The second article is the 11th chapter of the book, here published in the original English version. The German title of the chapter is: "Das Ovar nichtträchtiger und trächtiger Goodeiden" (engl.: The ovary of non-pregnant and pregnant Goodeids), and the article is from Mari Carmen Uribe et al., Laboratorio Biología de la Reproducción, Departamento de Biología Comparada. Facultad de Ciencias, Universidad Nacional Autónoma de México, Ciudad de México, México. We bring here both chapters in an unabridged version with all images, but changed for the current taxonomy. The third and last article is a publication of Uribe et al. from 2011: Ovarian Structure and Oogenesis of the Oviparous Goodeids Crenichthys baileyi (Gilbert, 1893) and Empetrichthys latos Miller, 1948 (Teleostei, Cyprinodontiformes). It deals exclusively with the situation of the egglaying subfamily Empetrichthyinae. We include this article here in a shortened version.
On the reproductive biology of Goodeids
1. What makes the reproduction of goodeids so remarkable?
When it comes to the reproduction of goodeids, the aquarium literature usually mentions two characteristics that make them stand out from the crowd of other aquarium fish: Mexican goodeids are "viviparous" (vívus, Latin = alive; párere, Latin = to give birth, to bring forth) i.e. viviparous. To be more precise, all matrotrophs are probably viviparous (máter, lat. = mother; trophé, gr. = nourishment), i.e. the young growing in the ovary (see below) not only cover their energy requirements via the yolk supply in the eggs (lecithotrophy; lékithos, gr. = yolk), but in the course of development the mother also contributes to the nourishment of the young. Splitfins have differently developed trophotaenia placentae (tainia, gr. = ribbon; placénta, lat. = cake), through which the growing embryo is supplied with food. Turner (1933) first listed these and other peculiarities of goodeid reproduction in a short article in the prestigious scientific journal "Science" and compared them with the conditions in the likewise viviparous poeciliids. He later documented this in more detail in a series of further papers. I will mention these peculiarities again here, even if some of the things Turner listed are seen somewhat differently today.
Turner (1933) has named a number of "characteristic complexes" that characterize goodeids with regard to their reproduction: Goodeids
1. have no "spermatophores", no "real" gonopodium, but some short fin rays on the anterior edge of the anal fin, have internal insemination and fertilization, but do not store sperm in the ovary,
2. have mature oocytes with little yolk, many of which degenerate when they reach their final size,
3. embryos spend only a short time in the follicles (follículus, lat. = sac, vesicle) of the ovary and, after most of the yolk has been consumed, begin to form special nutrient cords in the vicinity of the anus, which may differ in their organization in the various genera,
4. embryos increase significantly in volume in the ovarian cavity,
5. give birth to well-developed young. At the time of birth, the gonads are already recognizable as ovaries and testes. The nutrient strands are either shed or reabsorbed.
Individually, these characteristics are hardly unique to the goodeids. This also applies to the special features mentioned above, viviparity and the trophotaenial placenta. Many other bony fishes are also viviparous, including such popular aquarium fishes as poeciliids and hemirhamphids. Trophotaenia are also known from embryos of some viviparous Ophidiiformes ("visceral fishes"), some of which are found in the deep sea (e.g. Turner 1936; a detailed summary of viviparity in "fishes" and the various possibilities of "mother-child relationships" is given by Wourms et al., 1988). The "basic problems" associated with the reproductive mode "viviparity" are more or less similar in all viviparous organisms and are often solved in a surprisingly similar way, so that in the following I will sometimes take up results that have been worked out on other viviparous organisms, e.g. on poeciliids, which have been much better studied than goodeids, or hemirhamphids, if I am of the opinion that this could also apply to goodeids. Here, however, I will mainly refer to summaries (cf. Greven 1995, 2005, 2011).
2. Sex determination
All goodeids known to date are gonochoristic (goné, Greek = sex; choris, Greek = separate), i.e. there are males and females whose sex, as far as is known to date, is determined genetically ("genetic sex determination"). In some poeciliids (and other bony fishes), sex can also be influenced by natural environmental factors, especially temperature ("temperature-dependent sex determination"). If pregnant females of such species are kept at cooler temperatures, they give birth to more female offspring, while at higher temperatures more males are born. At temperatures in between, the sex ratio of the offspring is roughly balanced (summary in Greven, 2011).
The chromosomes of goodeids, as in the majority of teleosts, are very small. The double (diploid; diplóos, gr. = double) set of chromosomes, characteristic of somatic cells (sóma, gr. = body), is surprisingly variable and contains 24, 26, 28, 30, 42, 46 or 48 chromosomes, depending on the species, whereby the number of 48 seems to be the most common. However, two peculiarities should be mentioned: Females of Allodontichthys hubbsi have 42 chromosomes (2n), six of which are large and metacentric (metá, Greek = between; kéntron, Greek = center), i.e. they have two arms that are approximately the same length. Males, on the other hand, have 41 chromosomes with seven large metacentric chromosomes. This species therefore has several sex chromosomes. In general, a diploid set of 48 acrocentric (ákron, Greek = tip) chromosomes is considered to be original, but statements on relationships based solely on the karyotype (káryon, Greek = nucleus), i.e. the complete set of chromosomes and the appearance of the chromosomes, are very limited. Chromosomes are called acrocentric if their centromere (the point where the spindle "fibers" attach to the chromosome when the cell divides) is almost at the end of the chromosome. However, it has already been assumed on the basis of the karyotype that Ataeniobius toweri is not, as was assumed, a basal species, but is apparently related to the genus Goodea (summary in Uyeno et al., 1993).
3. Sex characteristics
Apart from clearly identifiable sex chromosomes, the sexes can also be recognized by specific, phenotypic (pha̅ínestai, Greek = to become visible) characteristics. A distinction must be made between primary and secondary sexual characteristics.
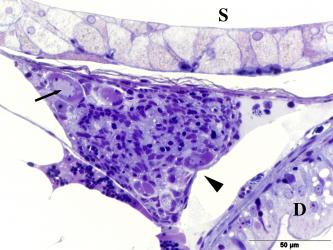
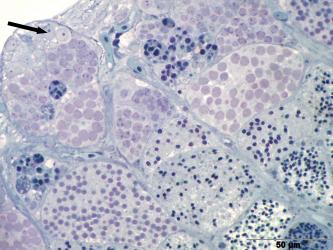
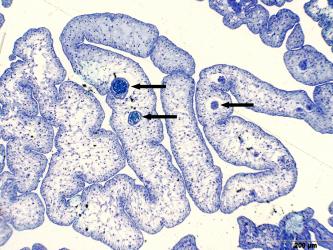
3.1 Primary sexual characteristics and gonadal ducts. Strictly speaking, primary sexual characteristics are only the gonads (= gonads), i.e. testicles and ovaries, but the gonadal ducts are often - not quite correctly - also included in the primary sexual characteristics. However, their development is obviously controlled by the sex hormones produced by the gonads. In principle, the gonads develop in the same way as in other bony fish (Guerrero-Estévez & Morena-Mendoza, 2012). The testes and ovary are paired during embryonic development (Fig.1). The paired anlagen approach each other in the course of gonadal development and finally fuse more or less completely. In the mature testis of goodeids, the two parts are fused in the anterior third, but then separate from each other (e.g. Kobelkowsky, 2005; cf. also Kingston, 1978, see Fig. 2). The mature ovary still has a septum, which divides the ovarian cavity into two more or less completely separate chambers (see Figs. 5, 6 and the excellent illustrations in Uribe et al. 2010 and in the article below). The time at which the testis can be recognized as a testis and the ovary as an ovary seems to vary depending on the species. Turner (1933) generally states that in goodeids testes and ovaries are already recognizable in newborns, but Mendoza (1937), for example, could not confirm this in newborns of large goodeids, e.g. Goodea atripinnis. In Chapalichthys encaustus, the testes and ovary can be distinguished from each other at around the time of birth (Guerrero-Estévez & Morena-Mendoza, 2012). Our own studies on "Xenotoca" eiseni show that the differentiation of the gonad already begins before birth, i.e. in the future testis precursor cells of the sperm, the primordial spermatogonia (= spermatogonia; spérma, gr. = the seed; gennán, gr. = to produce) and in the future ovary precursor cells of the mature oocytes, the primordial oocytes (= oogonia; oón, gr. = egg;) can be recognized (Fig. 1).
Left illustration (Fig. 1): A few days after birth, the originally paired gonade (arrow head) has coalesced into an unit. The big cell (little arrow) in the gonade anlage is an oogonium. „Xenotoca“ eiseni, histological preparation. D = gut, S = lumen of the swimbladder. Histological preparation (Greven & Pelzer, unpublished). Middle illustration (Fig.2): Transversal section through the posterior (conjugated) part of the testicle of „Xenotoca“ eiseni. The earliest stages of the spermiogenesis can be detected peripherally. In the efferenting ducts can be seen spermatozeugmae (arrow). Histological preparation. D = gut. Right illustration (Fig. 3): Cysts in the testicle of „Xenotoca“ eiseni showing different stages of spermiogenesis. Spermatogonia (arrow) are situated in the distal part of the lobule. Further explanations see text. Histological preparation.
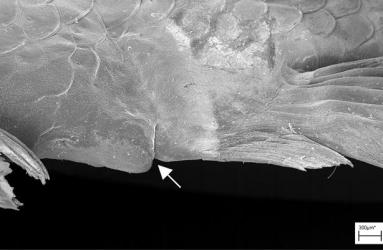
3.2 The testicles produce sperm and store sperm cells. In the mature testicle, the sperm are formed in lobules (lobulus = lobule, Latinized from lobós, gr. (= lobe). These are thin, blindly closed tubes at the edge of the testicle, which contain numerous spermatocysts (kýstis, gr. = bladder) (see Figs. 2 and 3). The spermatocyst (not to be confused with spermatocyte, see below) is formed by the germinal epithelium, which consists of somatic cells - called Sertoli cells in all vertebrates after the Italian physiologist and anatomist Enrico Sertoli (1842-1910) - and germ cells. Because the germinal epithelium does not fulfill all the criteria of an epithelium, the relationship between Sertoli cells and germ cells is also referred to as epithelial-like (epithelioid) (e.g. Grier et al., 2005). The youngest stages of the germ cells, the spermatogonia, are found where the lobules end blindly (Fig. 3). Further towards the center of the testis are spermatocysts, each with different stages of sperm formation, i.e. only one specific stage in each cyst, i.e. spermatocytes (kýtos, Greek = cell) of the first and second order, spermatids, which arise from the last maturation division (= meiosis), and mature sperm. The entire process of sperm maturation until the sperm is ready for fertilization is called spermatogenesis (génesis, Greek = formation). During meiosis (meion, Greek = less), the previously double set of chromosomes is reduced to half in two steps; the germ cells are then haploid (haplóos, Greek = single). The mature sperm (spermatocytes) are glued together in the cysts to form round sperm packets (= spermatozeugma or spermatozeugmata; zeugma, Greek = connection; singular: spermatozeugma), which are stored in the excretory duct system (see Fig. 2). A sexually mature male therefore always has a large quantity of spermatozoa "in stock". The excretory duct system consists of the two main ducts of the testis, the vasa deferéntia (vas, Latin = vessel; déferens, Latin = descending). These unite to form the unpaired sperm duct (= ductus spermáticus; dúctus, lat. = duct; spermáticus, lat. = belonging to the semen; cf. also Fig. 12), which opens into a space into which the contents of the urinary bladder are also emptied (= urogenital sinus or sinus urogenítalis; úron, gr. = urine; genitális, gr. = belonging to the genital organs; sínus, lat. = bay). Its orifice is visible directly at the attachment point of the modified anal fin (Fig. 4). The individual spermatozoon is relatively small; it has a head (in which the genetic material is condensed), a middle section that essentially contains mitochondria, which provide the energy for movement, and a relatively long tail, the flagellum, with the help of which the spermatozoon moves. An acrosome (ákron gr. = tip; sóma, gr. = body) at the front end of the sperm, which is characteristic of most vertebrate sperm and contains enzymes that locally dissolve the egg membrane so that the sperm can penetrate the egg, does not exist in the vast majority of bony fishes, including the goodeids. So far, the sperm of only very few goodeid species have been studied in detail. Although they are unlikely to differ from each other in terms of the basic organization (head, midpiece, tail), there may be differences in size and other characteristics, e.g. those that influence motility and swimming speed. The spermatozoa of the goodeids differ clearly from the sperm packets of the poeciliids. In the latter, the sperm nuclei are on the outside of a spermatozeugme, whereas in the goodeids they are clearly on the inside. Even a single spermatozyme is likely to contain many more sperm - certainly thousands - than are necessary for insemination and fertilization of the eggs for a litter. Surprisingly, more precise information on the number of sperm per spermatozygote and how many spermatozygotes are actually transferred during copulation is not yet available, but sperm counts after collecting the spermatozygotes (possibly by stripping the males) in a blood counting chamber - as is common in poeciliids (e.g. Kuckuck & Greven, 1997) - should not be a major problem. If it is possible to obtain spermatozoa from an anaesthetized male, e.g. by carefully massaging the flanks, artificial insemination should also be possible in goodeids if appropriate care is taken, by using the finest glass capillaries to introduce suspensions of sperm to or into the relatively small female genital opening (cf. Fig. 10).
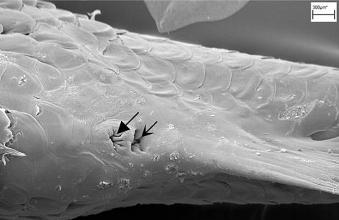
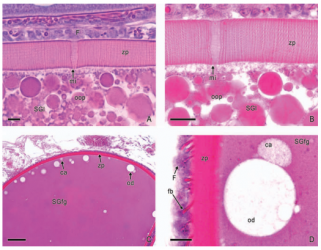
Left illustration (Fig. 4): The urogenital cleft of the male is a transversal slit (arrow) in front of the insertion of the anal fin. „Xenotoca“ eiseni, scanning electron microscope picture (ex Greven & Brenner, 2010). Middle illustration (Fig. 5): Transversal sections of the ovaries of different Goodeids, showing the position of the follicles (black dots). Top down: Xenotoca variata, Xenoophorus captivus, Neotoca bilineata, Ataeniobius toweri (ex Hubbs & Turner, 1939, modified). Right illustration (Fig. 6): Septum in the ovary of a female of Ameca splendens. Histological preparation. Blood vessels (arrows).
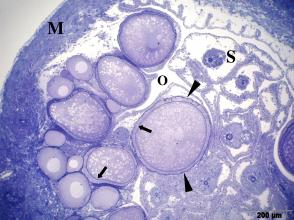
3.3 The ovary forms eggs with a low yolk content. The mature female ovary is a hollow organ whose lumen (lúmen, Latin = cavity) is divided by a longitudinal septum (Figs. 5 and 6). The mature ovary is covered on the outside by a thin layer of cells (peritoneal epithelium), which is followed by a layer of connective tissue with muscles and blood vessels. The ovarian cavity is lined by a layer of coherent cells (ovarian epithelium), which in turn contains germ cells and somatic cells (also referred to in its entirety as germinal epithelium), as in the testis. Germ cells and some of the somatic cells are transferred to the connective tissue (stróma, Greek = storage site) of the ovary, where the somatic cells become the "wall" of the follicle, consisting of only one cell layer, in which the young egg cell (oocyte) is located (see Mendoza, 1943, Grier et al., 2005, Uribe et al., 2005, Uribe et al., 2010). Depending on the genus, the follicles may be restricted to certain areas within the ovary (Fig. 5 and 7). A distinction is essentially made between ovaries in which the follicles are located in two dorsolateral folds to the side of the septum and ovaries in which the follicles are located in the ovary wall and the septum (Hubbs & Turner, 1939, Mendoza, 1956). During oogenesis, the oogonia grow, become first and second order oocytes, analogous to sperm development (see above), and are haploid after meiosis. During this time, the yolk is also stored (= vitellogenesis; vitéllus, lat. = yolk), which the mother provides via precursors called vitellogenins, which are formed in the liver. These vitellogenins are relatively large protein molecules that reach a mass of 348 kDa (= dalton; unit of mass of atoms and molecules) in goodeids (Vega- López et al., 2006) and therefore cannot easily enter a cell. The amount of yolk in the eggs of goodeids is reduced in comparison to the amount of yolk present in egg-laying Cyprinodontiformes; the eggs are therefore relatively small and are considered to be low in yolk (see also Turner, 1933, Mendoza, 1940). One might think that the production of vitellogenin would cease with the completion of egg maturation. However, this is not always the case in goodeids. In Girardinichthys viviparus, vitellogenins are also produced during gestation, which then apparently benefit the growing young; in Ameca splendens, they are stored in the fatty tissue beforehand for this purpose (Vega-López et al., 2007). The developing oocyte is surrounded by an envelope, which is formed by the oocyte itself, but in some cases probably also by the follicular epithelium (Riehl & Greven, 1993). This envelope is given very different names in the literature, e.g. chorion (Greek = envelope) or zona radiata. The "radiata" (radiatus, lat. = radiating, provided with rays) refers to the fact that during its formation the egg membrane is formed between fine protrusions of the egg cell, called microvilli (singular: microvillus; mikrós, gr. = small; villus, lat. = villus), which initially gives it a striped appearance (under the light microscope), provided it is thick enough. Nowadays, however, the term "zona pellucida" (zóna, gr. = belt, zone; pellúcidus; lat. = transparent) seems to have become generally accepted for the egg membrane of fish, because it resembles the egg membrane of mammals in some (molecular) details. This zona pellucida is, as in all livebearers, extremely thin and only 0.5-1.2 µm thick in mature goodeid oocytes. Therefore, a "striation" is not visible in the light microscopic image (Fig. 7). Perhaps the thin thickness of the egg membrane was a necessary precondition for the development of viviparity or a consequence of this mode of reproduction. A thin egg membrane certainly contributes to the facilitation of gas exchange between maternal and embryonic tissue (cf. Riehl & Greven, 1993; Greven, 1995). Another special feature - and to my knowledge this applies to all viviparous bony fishes - is the fact that no micropyle (pylé, Greek = gate) has yet been found in the zona pellucida of the fertilization-ready eggs, i.e. a tiny opening that allows the sperm, which has no acrosome to enter the egg cell (e.g. Gravemeier & Greven, 2006). It is therefore still unclear how a sperm enters the mature egg. However, there is often a depression in the ovarian epithelium above the oocyte (which is also sometimes referred to as a "delle" in Anglo-American literature), in which sperm can be found after insemination (Mendoza, 1943), but apparently also for some time afterwards (see Uribe et al., 2005, Chapter 9). This dent is the site through which the sperm enters the egg (see the discussion in Greven, 2011). It is also the site through which the embryo enters the ovarian lumen (Mendoza, 1943). Since the mature oocytes of the goodeids are low in yolk, the lecithotrophic phase of the young, i.e. the period in which they feed on the yolk, is inevitably short. There is very little comparative data on the size of oocytes ready for fertilization. Turner (1933) gives a diameter of only 0.3-0.5 mm for mature oocytes of Goodea bilineata. Uribe et al. (2005, Chapter 9) mention diameters of 0.8-1.3 mm for Goodea species and 0.8-1 mm for Ilyodon whitei, Girardinichthys viviparus and Characodon lateralis. Overall, such data can already provide clues as to whether intensive or less intensive feeding by the mother can be expected. A considerable number of the follicles in the ovary become atretic (a, gr. = generally denotes an absence; trésis, gr. = opening), i.e. the follicles degenerate. This is a completely natural process that can affect oocytes before and after yolk storage. The atresia is therefore not cyclical (Mendoza, 1941). In pregnant females, mainly non-inseminated mature eggs degenerate at the beginning of gestation and towards the end of gestation also eggs in an early stage of yolk storage (Neotoca bilineata: Mendoza, 1941; Ilyodon whitei, Goodea atripinnis: Uribe et al., 2006). Turner (1933) was of the opinion that such atretic follicles would ultimately benefit the young growing in the ovarian cavity (cf. also Mendoza, 1943). The two halves of the ovary open into a short unpaired gonoduct (goné, Greek = sex; ductus, Latin = duct). This opens between the anus and the base of the anal fin, which is not particularly modified in females (Fig. 8), in the immediate vicinity of the ureteral orifice, so that there is no clear urogenital sinus (see Kobelkowsky, 2005). A receptaculum seminis (receptáculum, lat. = receptacle, pouch; sémen, lat. = semen), in which sperm are stored long-term, does not exist in goodeids (e.g. Mendoza, 1941, 1962).
Left illustration (Fig. 7): Ovary of Ameca splendens. Oocytes in different stages of development. Histological preparation. Embryonic membrane (arrow), follicle epithelium (arrow head); M = Muscle layer. O = Ovarian lumen. S = Septum. Middle illustration (Fig. 8): Genital opening (slim arrow head) of a female of „Xenotoca“ eiseni between insertion of anal fin (right) and anus (thick arrow head). Scanning electron microscope picture (ex Greven & Brenner, 2010). Right illustration (Fig. 9): Female (above) and male (below) of Girardinichthys multiradiatus. Please note the sexual dimorphism in fins. Copyright: Markus Heußen
3.4 Secondary sexual characteristics and sexual dimorphism. Males and females naturally differ in numerous other characteristics, the formation of which (as well as the formation of the above-mentioned excretory ducts) depends on sex hormones (testosterone, oestrogens), which are produced by special cells in the gonads. Sexual dimorphisms in size and coloration are common in goodeids, although there are also species whose sexes do not or hardly differ in color. The males are usually smaller than the females, or both sexes are about the same size or may have a differently shaped dorsal fin, as in Skiffia species, for example (Mendoza, 1965). Sexual dimorphism is particularly striking in Girardinichthys species (Fig. 11; e.g. Macías-García, 1994, Domínguez-Domínguez et al., 2005). The evolution of conspicuous sex differences is essentially due to the fact that males compete with each other for females, and that females preferentially select certain characteristics of males ("female choice"). The two are not necessarily mutually exclusive. The production and maintenance of such ornaments (also) indicating the genetic quality of a male - meaning not only color patterns, but also larger fins or other conspicuous features - are a costly affair, which are preferred by females because they indicate a certain quality of the male in question to the female, but are unfortunately also conspicuous to possible predators (e.g. Macías-García, 2010). Macías-García et al. (1994) have shown in laboratory experiments that the females of Girardinichthys multiradiatus prefer males with larger fins, but that these males are also more likely to be preyed upon by a natural enemy of these fish, the garter snake Thamnophis melanogaster, than males with smaller fins. The larger fins may also contribute to a reduction in mobility. This can lead to an overall reduction of attractive males in a population (Macías-García et al., 1998). Garter snakes are also more likely to notice the speckled males of the geographically very variable species Xenotoca variata than the less speckled ones (Moyaho et al., 2004). Two other secondary sexual characteristics of goodeid males are more directly related to reproduction. They have become known under the not entirely fortunate names "pseudopenis" and "andropodium".
3.5 "Pseudopenis" or "urogenital organ" of males
The above-mentioned unpaired spermatic duct of the testis and the ureter are surrounded by special muscles, which are apparently there to push the spermatozoa out of the sperm duct during mating. The whole structure has therefore been called a pseudopenis (pseudo-, Greek = false) (Mohsen, 1961a) or neutrally a urogenital organ (Nelson, 1975). This organ has only been histologically examined in about a dozen goodeid species. In Goodea atripinnis, the musculature of the urogenital organ consists of three muscle bundles, of which a relatively thick bundle lies ventrally and runs transversely to the longitudinal axis of the fish (see also Fig. 10). In addition, there is a longitudinal bundle on each side. Ductus spermaticus and ureter are embedded in this musculature at different depths in the course of the elongated organ. In Skiffia lermae it is said to be a single "muscle mass", likewise in Characodon lateralis, but here it is said to have originated from the fusion of three muscle bundles (Mohsen, 1961a, Nelson, 1975). The previous descriptions of the urogenital organ still leave many questions unanswered. Its development and its apparent structural diversity should be investigated comparatively in an even larger number of goodeid species.
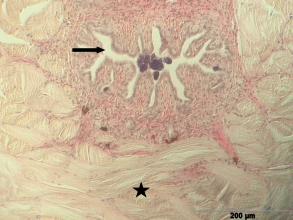
Left illustration (Fig. 10): Ductus spermaticus (arrow) with spermatozeugmae. Below across body axis running muscles (star) of the urogenital organ of „Xenotoca“ eiseni. Histological preparation. Middle and right illustrations (Fig. 11): Anal fin of a female (left) and male (right) of „Xenotoca“ eiseni. Please note the shortened first fin rays of the male. Cleared and with Alizarin stained preparations
3.6 The anal fin of males: "gonopodium", "andropodium" or "spermatopodium"?
The anal fin of males (in contrast to the anal fin of females) is clearly divided into two lobes. It is already visible to the naked eye that their first rays are shortened. This becomes clearer in lightened specimens in which the bones have been stained red (Fig. 11). Ray 1 remains small from the beginning in both sexes (see Fig. 14). Turner (1933a) associates this modified anal fin with reproduction, but only says, alluding to the conditions in poeciliids, that this is not a "true" gonopodium. However, this did not prevent later investigators and Turner himself from referring to the anal fin of goodeid males as a gonopodium (goné, gr. = procreation; pus, podós, gr, = foot) (e.g. Turner et al., 1962, Miller & Fitzsimons, 1971). Other authors also call them andropodium (anér, andrós, gr. man) (e.g. Meyer & Lydeard, 1993, Hieronimus, 1995), often also spermatopodium (Mohsen, 1961 and in numerous scientific publications) or more recently even abdomopodium (Meyer et al., 2001). Meisner (2005) has suggested - not very consistently - that all modifications in male "fish" that have something to do with insemination should be called "intromittent organ", even if they are not inserted into the female's sexual opening. Apart from the fact that these different designations are not very informative, a special name for the modified anal fin of the goodeids is not absolutely necessary if one considers its actual function during mating. Turner et al. (1962) studied the development of the anal fin of males in "Xenotoca" eiseni and also compared the anal fins of adult males of different species. The anal fin of newborn "Xenotoca" eiseni has 15 (sometimes 16) fin rays. In males, rays 2 to 7 of these remain behind in the course of development. However, this only becomes fully apparent 78 days after birth. In some species, e.g. Ataeniobius toweri or Goodea atripinnis, some of these fin rays are even bifurcated. Such a bifurcation of the fin rays seems to be the norm in females. In general, the study shows that the anal fin of goodeid males can vary so much - Turner et al. (1962) differentiate between specialized and less specialized anal fins - despite their seemingly uniform structure that it can probably be used to differentiate species or at least genera (Fig. 12). How these changes correlate with the differentiation of the testis and the concentration of sex hormones, which control the formation of the anal fin, has not been investigated. The anal fin of the male is connected to the vertebral column via various bones, which in their entirety are referred to as the suspensorium (suspéndere, Latin = to hang up) (e.g. Guzmán, 1994, Díaz-Pardo, 2005). The suspensorium consists of three regions, a dorsal, a middle and a ventral one. The dorsal region consists of the vertebrae located in front of the tail (= precaudal vertebrae) and the first caudal vertebrae, the middle region consists of elongated bony elements (the bony fin supports [= radialia] located closer to the center of the body [proximal], which are usually connected to the hemal processes of the vertebrae, and the ventral region consists of small [distal] radialia located further away from the center of the body [distal], which are connected to the fin rays of the anal fin). Within the Girardinichthynae, for example, the number of vertebrae (7-16) and proximal radialia (13-23) involved and their length vary depending on the species, so that the suspensorium can also be used to diagnose the species (Díaz-Pardo, 2005 and Fig. 13).
Left and middle illustrations (Fig. 12): Different shapes of the first 8 rays of the anal fin of males of Xenotoca variata (left) und Goodea atripinnis (right) (ex Turner et al., 1962, modified). Right illustration (Fig. 13): Organisation of the suspension in different species of the Girardinichthyini. The number of involved vertebrae (in this case Nr. XV–XXII) and proximal radialia is differing from species to species (ex Díaz-Pardo, 2005, modified).
4. When do goodeids become sexually mature and when and how often do they reproduce?
Apparently, there is little really accurate data available on when and under what conditions the various goodeid species become sexually mature in the field and in the aquarium, whether there are differences between females and males and when the animals become sexually active for the first time. I will therefore only give a few examples (see also the compilation in Hieronimus, 1995 and the website of the Goodeid Working Group). In general, it will probably be the case that the males - as observed by Kempkes (1998) in Zoogoneticus tequila and as also known from poeciliids - already court and attempt to copulate before reaching sexual maturity. Zoogoneticus tequila becomes sexually mature under laboratory conditions at temperatures between 26 and 28 °C at the age of 6-10 weeks (Arbuatti et al., 2011). In the field, Allotoca diazi should reach sexual maturity within a year, the large species Alloophorus robustus and Goodea atripinnis only in the second year (Mendoza, 1962). After reaching sexual maturity, the males repeatedly produce mature sperm from the spermatogonia lying ready in the testes and the females produce mature eggs from the oogonia lying ready in the ovary. These processes are in part strongly dependent on the climate (especially the temperature) and the length of the day, so that egg maturation in particular can be reduced or even stopped completely in colder months. There are also species that give birth several times a year, such as Girardinichthys viviparus mainly between March and October (Díaz-Pardo & Ortiz-Jiménez, 1985), Ameca splendens with a maximum of births in March to May and in September to November (Ortiz-Ordónez et al., 2007), Allotoca diazi from April to February of the following year, "Xenotoca" eiseni and others, or very probably only once like Alloophorus robustus and Goodea atripinnis from June to August (Mendoza, 1962). Such cycles are apparently maintained for some time in the aquarium (Hieronimus, 1995). It is also important to know when eggs are ready for fertilization. There are relatively few recent studies on this. In Ameca splendens, the oocytes develop asynchronously, which means that females probably also have eggs that are ready for fertilization shortly before the birth of the young (Ortiz-Ordónez et al., 2007). In Neotoca bilineata and Quintana atrizona, too, egg production does not come to a complete standstill during gestation and egg maturation does not occur abruptly shortly before the birth of the young (Mendoza, 1941).
5. Reproductive behavior
5.1 Mate choice
Goodeid males mate. Various things can be deduced from this fact alone. There is no doubt that females recognize males of their own species and males recognize females of their own species by means of visual (visible) and chemical signals, just like other "fish". Nothing is (yet) known about the latter in goodeids. However, it can be assumed that the females release attractant substances. In egg-laying and live-bearing fish, these can be steroid compounds or prostaglandins (a group of tissue hormones) from the urine, for example (summary in Greven, 2011, among others). Visible signals, e.g. striking colors or color patterns, extravagant structures such as enlarged fins and/or characteristics during courtship (intensity of courtship, etc.) allow the female to assess the quality of the partner and select the "best" one (e.g. Greven, 1999, see also Macías-Garcia & Valero, 2010). Many goodeids show a clear sexual dimorphism in size, finning and color pattern. The males are often more conspicuously colored in the spectrum of light visible to us. In addition, there are also signals in the short-wave range of light (UV range 300-400 nm) that cannot be perceived by us. Macías-Garcia & Burt de Perera (2002) have shown that females of Girardinichthys multiradiatus, for example, can perceive UV light and that UV-reflecting patterns on the body of males are important when choosing a mating partner. The size of the sexual partners raises interesting questions. A preference for larger males by females seems to be typical for poeciliids, for example. However, the tendency to display increases with body size, so that it is not always clear whether size or behavior is preferred. In the case of "Xenotoca" eiseni, experiments have shown that - perhaps for mechanical reasons - both males and females prefer a sexual partner of approximately the same size (this is referred to in Anglo-American literature as "size assortative mating"), as this is likely to result in better coordination in the transfer of spermatozoa after courtship (Bisazza, 1997). In the wild, too, mating partners of the same size are apparently preferred (Girardinichthys multiradiatus: Macías-Garcia, 1994). However, this does not prevent smaller males, e.g. of "Xenotoca" eiseni, from constantly harassing larger females and attempting copulations (cf. Greven & Brenner, 2010) or preferring large females in choice attempts (Greven & Kleinhammans, unpubl.).
Fig. 14: Mating of „Xenotoca“ eiseni, from below; after 2200 ms the pair is separating. Last picture: Pair during copulation. From Video-sequences (ex Greven & Brenner, 2010). Photo 1: 0 ms; Photo 2: 440 ms; Photo 3 720 ms; Photo 4: 1400 ms; Photo 5: 2200 ms; Photo 6?
5.2 Courtship
Courtship processes are described in more or less detail in scientific (e.g. for Goodea atripinnis: Nelson, 1975; Characadon lateralis and various Xenotoca species: Fitzsimons, 1972, 1976; Bisazza, 1997), Girardinichthys viviparus: Díaz-Pardo & Ortiz-Jiménez, 1985; Macías-Garcia, 1994; Zoogoneticus tequila: Arbuatti et al., 2011) as well as in the aquaristic literature (see also the compilation in Hieronimus, 1995). A comparative analysis and a standardized terminology for the individual elements shown during courtship (which are certainly not yet fully recorded in the various species) are, to my knowledge, still lacking. Macías-Garcia (1994) distinguishes between male-female fin display, courtship fin folding, parallel swimming, overtaking, flagging, a figure-of-eight dance, vibrating, and copulation attempts and copulation in Girardinichthys multiradiatus, flagging, a dance in which the figure-of-eight dance is described, vibrating as well as copulation attempts and copulations, without describing these behaviors in detail and in the context of the entire courtship. Zoogoneticus tequila is one of the species with an apparently quite simple courtship display. The male actively searches for females, then swims next to the female for a long time, presenting mainly the yellow stripes of his caudal fin with rapid body movements - although this does not necessarily precede mating - and stands next to the female at an angle of about 45° before copulation. Pregnant females are obviously not stalked. Courtship does not begin again until 1-2 days after the birth of the young (Arbuatti et al., 2012). Kelley et al. (2005) report that males of Skiffia multipunctata constantly court females. The male brings himself in front of or slightly to the side of the female and wiggles his dorsal and anal fins, positions himself, head or tail down, in front of the female and performs 180° turns. Males of Ameca splendens stand upside down in front of the female, shake their front body violently and sometimes turn 180° to present the other side of their body as well (Kelley et al., 2006). Fitzsimons (1972, 1976) and Nelson (1975) still provide the most detailed information. Nelson (1975) also provided an exact description of copulation. Fitzsimons (1972) distinguishes six elements in the courtship display of males of Characodon lateralis, namely lateral T-formation, with which the male stops a swimming female; lateral tail beating; oblique display to the right or left of the female, whereby the male turns his caudal fin and caudal peduncle towards the female; lateral head-down and lateral head-up next to the female combined with head shaking), in the courtship display of "Xenotoca" eiseni five elements, again the T-position, sometimes combined with trembling of the male; swimming towards the female from behind and right-left movements in front of the female ("lateral wheeling"); dancing above the female in loops resembling a figure eight ("loop dance"), half dance, and standing at an angle. Males of different populations of "Xenotoca" eiseni can differ in their courtship behavior. In other Xenotoca species, other elements can also be distinguished, e.g. in "Xenotoca" melanosoma a zig-zag dance and head wagging. For further details see Fitzsimons (1972, 1978). We observed the courtship of an aquarium strain of "Xenotoca" eiseni a long time ago and never saw any half-dances or pronounced figure-eights, so we neutrally described the "loop dance" as a change of sides, the T-formation as locking, the "lateral wheeling" as swimming from the lurking position and standing at an angle as a lurking position. Cooperative females remained on the spot or sank to the ground trembling. Some females clearly shook their heads when a male approached them, but not to invite it to court (see Hieronimus, 1995), but to ward it off. According to some authors, females are only receptive (recípere, lat. = to receive) shortly after giving birth and males are not able to force copulation (see Macías-Garcia & Valero, 2010). The courtship of Goodea atripinnis is also relatively complex. Nelson (1975) distinguishes three phases: In the orientation phase, the male observes and follows the female. The subsequent display includes several elements (sigmoid position; C-curvature so that the head and tail point towards the immobile female; lowering of the head in front of the female as well as trembling, fin flapping and swimming forwards and backwards; swimming in a tight circle or semi-circle in a C-shape; round dance in which the male swims around the female and copulation). If the female is receptive (and cooperative; for terminology, see Greven, 2005), she wags her head (this promotes the intensity of the courtship) and fades as the courtship progresses. A non-receptive (non-cooperative) female will flee from a courting male or occasionally become aggressive. These few examples show that there are obviously a number of similarities in the courtship displays of different goodeids, but also clear differences, so that it is of course still of interest to carefully record various courtship displays and, if possible, to document them photographically. Males of many goodeids are territorial, i.e. they defend an area against other males and mate with the females here. This is also evident in goodeids in the aquarium (Hieronimus, 1995, Kempkes, 2002). Macías-Garcia (1994) was able to show in field studies that the males of the sexually dimorphic species Girardinichthys multiradiatus defend areas near the sun-warmed banks of water bodies that are frequently visited by females. The males approach any conspecific that swims into such an arena. Females are stalked, males fight each other. The reason for these fights is therefore the females and the water areas in which they are present. As the females can (only) be inseminated shortly after they have given birth, it may pay off for the males to defend areas that are frequented by females shortly before they give birth. This leads to an operational sex ratio shifted in favor of the males in these water zones. The author even calls these areas "courtship arenas" and compares the whole thing to so-called lek systems, in which the males present themselves to the females (the best-known example of this is the ruff, Philomachus pugnax) and the females choose a suitable partner from these. In Girardinichthys multiradiatus, however, these "arenas" are a "territory" to be defended by the males and at the same time places where the females deposit their young. It is very unlikely that this can be observed in the cramped conditions of an aquarium. Studies in outdoor facilities might be a good idea here.
5.3 Copulation: how and with what?
The modified anal fin is only indirectly involved in copulation, i.e. the direct act in which the male places his spermatozoa in the female's genital opening. Many statements in the aquaristic literature as well as the term gonodpodium etc. give the impression that the anal fin or parts of it can be inserted into the female genital opening. This is not the case; therefore the term "intromittent organ" is also misleading (see above). The most detailed description of copulation can be found in Nelson (1975). After courtship, the male slides to the side of the female, curves his anal region towards the anal region of the female and tilts his anal fin in the latter's direction. The male then surrounds the female's genital opening with the small anterior lobus of his anal fin; the incision of his anal fin comes into contact with the anterior edge of the female's anal fin. In this way, a pouch is formed, the sides and bottom of which are formed by the anterior lobe of the male's anal fin. Its roof is the postanal region of the female with the genital opening. Caudally, the pouch is closed by the anterior edge of the female's anal fin. The male's genital opening is then close to the opening of this pouch. The whole process takes place with trembling movements and lasts up to five seconds. Nelson's observations can also be largely confirmed for smaller species, e.g. "Xenotoca" eiseni. In this species, the male approaches the female from the side after courtship (locking, changing sides, see above), swings the anal fin towards the female's genital pore and wraps his caudal fin around the female's hind body from below (see Fig. 14; Greven & Brenner, 2010; Greven & Kleinhammans, unpublished). The separation of the partners is usually violent and very fast. During copulation, the muscles of the urogenital organ apparently contract, so that the spermatozoa or the spermatozoa are literally "shot" via the pouch into or at (?) the female's genital opening. A part of the anal fin is therefore rather one of several aids that guide the spermatozoa in the right direction and thus prevent spermatozoa from being lost during the copulation process. However, such a function of the anal fin of males is not limited to goodeids, but is also known from egg-laying fish with external insemination and fertilization (cf. Greven & Brenner, 2010). Nevertheless, goodeid males with this rather inadequate equipment also attempt to mate with non-cooperative females, i.e. to rape them in the truest sense of the word. We have often been able to observe in "Xenotoca" eiseni how a fleeing, often much larger female was pursued by a male and repeatedly attempted to copulate (see also Greven & Brenner, 2010). Since a high degree of synchronization between male and female is apparently required for successful copulation (see Bisazza, 1997), we do not know whether such rape attempts can also be successful. Macías-Garcia & Valero (2010) are even of the opinion that rape ("force copulation") is not possible in goodeids. Perhaps the whole thing can only be classified as sexual harassment, which is also very intense in goodeids (Macías-Garcia & Valero, 2010).
If sperm cells have been transferred to the female, they must be dissolved in the female genital tract so that the sperm can migrate into the ovary. Some of the sperm then apparently collect in the above-mentioned dents above the egg cells. If the eggs are already inseminated and fertilized in the follicle, the sperm must overcome the ovarian epithelium (in this case the "floor" of the dent), the underlying connective tissue, the follicular epithelium and the thin egg membrane. How this happens is unknown for all viviparous teleosts. Excess sperm are probably removed from the cell layer (epithelium) that lines the ovarian cavity, as is apparently the case in poeciliids (see summary in Greven, 2011). However, corresponding studies on goodeids are lacking. Multiple paternity has been studied in detail in poeciliids, especially in guppies (summary in Evans et al., 2011). It is also to be expected in goodeids that several males are the fathers of the young of a litter. In laboratory experiments with Girardinichthys multiradiatus, two males of different sizes were allowed to mate with one female. The offspring came from both males, but the males that mated with the female first had the greater reproductive success (Macías-Garcia & Saborío, 2004).
6. Mother-child relationships
6.1 What is matrotrophic viviparity?
The term "matrotroph" expresses the fact that the young growing in the ovary are fed by the mother. However, the fact that newborn goodeids are relatively large is not necessarily a sign of such a food transfer. This increase in mass could be due to the fact that the eggs have stored so much yolk that it is sufficient for the entire embryonic development (as is the case with the guppy Poecilia reticulata, for example) and/or that the increase in mass is primarily due to fluid retention. Initial indications as to whether a lecithotrophic or matrotrophic viviparity is involved are provided by dry weight determinations of eggs and various developmental stages of the young up to birth. A strong weight loss in the newborns speaks for lecithotrophic (lékithos, Greek = yolk) viviparity, which obviously does not occur in goodeids, although the embryos have a short lecithotrophic phase at the beginning of development due to the low yolk supply. Only a slight weight loss, but above all a constant weight or a strong weight gain, indicate a food transfer from mother to child, i.e. matrotrophic viviparity. Such weight gains have been determined in some goodeids. Depending on the species and the developmental stages studied, the dry weight increases by a factor of 10 to 387 (Wourms et al., 1988; Hollenberg & Wourms, 1995). The example of Ataeniobius toweri shows that the trophotaenia do not always have to play the main role in the nutrition of the young, whose newborns have a considerable size (so that matrotrophy can initially be assumed, cf. also Turner, 1940), but which can only absorb food via their trophotaenial rudiments at the beginning of development - if at all. More detailed studies would be of interest here.
6.2 Follicular or ovarian (intraluminal) gestation?
It is repeatedly emphasized that the mature oocytes of goodeids (as well as those of poeciliids and hemirhamphids) are already fertilized in the follicle. After careful examination of numerous Neotoca bilineata, Mendoza (1943) came to the conclusion that fertilization, completion of meiosis and the time of ovulation (the moment when the eggs or embryos leave the follicle and enter the ovary; ovulátio, Latin = egg-laying) occur almost simultaneously and very quickly. He practically never found fertilized eggs in the follicle, but only in the ovarian cavity. In addition, he found (at least for Neotoca bilineata) that the mature eggs are only fertilized about seven days after mating. This probably correlates with the change in coloration of the females. It is probably for these reasons that the information on the stage at which the developing embryos are released into the ovarian cavity is so imprecise. Turner (1933a) states that the embryos are released into the ovarian cavity shortly after fertilization of the eggs, but also that the yolk is still resorbed in the follicle and the trophota begin to form. In a later work he writes that this occurs during the first stages of division of the fertilized egg (Turner, 1940). Miller & Fitzsimons (1971) note that the embryos of Ameca splendens remain in the follicle until the yolk is consumed, while Mendoza (1958) is of the opinion that the eggs are released into the ovarian cavity at the time of fertilization. Uribe et al. (2005; Figure 8) imaged very young germs (germinal disk: stage in which the cells of the germ as a disk, on the yolk-containing part of the egg cell that is not involved in the cell divisions) in the ovary cavity of Ilyodon whitei and reported that they had found embryos in this and in the neurula stage (stage in which the neural tube is formed) in the ovary cavity of Goodea atripinnis (s. a. Uribe et al., 2010), i.e. in a developmental stage that is significantly younger than embryos with incipient trophotaenia formation and also still has a relatively large amount of yolk. At this stage, the embryo is also still surrounded by the thin egg membrane. The strong muscle layer of the ovarian wall probably plays a role in the ejection of the embryos into the ovarian cavity, as such early stages of development cannot free themselves from the follicle on their own. I would not be surprised if there were also differences in the timing of ovulation between the various species. What is certain is that the young do not (as in poeciliids) remain in the follicle until birth (= follicular gestation), but spend most of their development in the ovarian cavity (= intraluminal gestation). Wherever a young animal grows up "in" the mother or "in" the father, it causes its parent (there is such a word!) problems, which are all the greater the more the adolescents depend on him. A general problem is that the embryo contains foreign antigens for the mother (or father), which should lead to the formation of antibodies. The embryos should therefore actually be rejected. However, the fact that this does not happen is probably due to properties of the epithelium lining the ovarian cavity, due to which the ovarian cavity is literally sealed off from the underlying connective tissue (in poeciliids this would be the follicular epithelium; see Greven, 2011). However, this has not been investigated in goodeids.
6.3 Superfetation with goodeids?
Superfetation (super, lat. = over; fétus, lat. = body fruit) is characterized by the fact that embryos of different developmental stages are present in the ovary of a pregnant female at the same time, as the more or less continuously growing oocytes are also fertilized at different times. The birth of the young, which gradually grow up, therefore takes place over a longer period of time. Superfetation is known in a number of hemirhamphids and poeciliids, among others. A prime example of superfetation in poeciliids is the dwarf molly Heterandria formosa; here - as in the vast majority of cases - it is correlated with matrotrophic viviparity (see above). Turner (1940) reported superfetation "in rudiments" in various goodeids (including Xenoophorus captivus, Chapalichthys encaustus, Zoogoneticus quitzeoensis, Skiffia lermae, Neotoca bilineata, Characodon lateralis), but at the same time drew attention to the decisive differences to the apparently very successful superfetation of some poeciliids. In the latter, each embryo remains in a follicle until birth, where it can be individually cared for to a certain extent, whereas in goodeids the embryos probably reach the ovarian cavity relatively early, so that those from the first "insemination wave" (i.e. the larger number of young in each case) are much further developed than those that have arisen from the possibly later fertilized oocytes. Turner (1940) was only ever able to observe very early stages among the young of a litter in the ovary, but never older ones, and he only found degenerating ones of the middle developmental stages. The "stragglers" obviously do not survive in the environment in which their older siblings grow up, so such superfetation would be very ineffective. However, such phenomena presuppose, among other things, that oocytes also mature during a pregnancy (this should be the case in species with asynchronous development; cf. also the discussion in Turner, 1940) and that viable sperm ready for fertilization are still or again present in the ovary, i.e. sperm would have to survive in the ovary cavity for a longer period of time or pregnant females would have to allow themselves to be mated again. These observations should urgently be investigated and documented more convincingly. Normally the newborns are about the same size and equally developed (see also Mendoza, 1937). Díaz-Pardo & Ortiz-Jiménez (1985) found three individuals among 128 females of Giradinichthys viviparus that had embryos of different lengths in the ovarian lumen. In one case, the largest embryo (of several) was three times larger than the smallest (of several). The authors refer to this as superfetation and believe that this type of reproduction occasionally occurs in Giradinichthys viviparus and that superfetation is not abnormal in goodeids, as Mendoza (1941, 1962) generally suspects. Some time ago we also repeatedly found juveniles in the ovary cavity of Ilyodon furcidens which differed considerably in size. Unfortunately, we did not check at that time whether these were perhaps only fledglings of one and the same developmental stage (which would be conceivable, as the young depend heavily on having access to maternal resources via the trophotaenia after the short lecithotrophic phase) or actually different developmental stages. In any case, the discussion about possible superfetation in goodeids is not yet over. However, goodeid breeders could start by recording at what intervals and in what size the young are born.
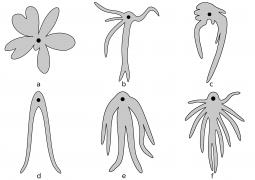
Left illustration (Fig. 15): Different types of trophotaeniae (schematic); a Goodea atripinnis; b and c Xenoophorus captivus; d Characodon lateralis; e Xenotoca variata; f Zoogoneticus quitzeoensis. Black circle = Anus (ex Hubbs & Turner, 1939, modified). Middle illustration (Fig. 16): Development of the trophotaenial placenta. Cells of the gut are grey, cells of the body surface black marked. The arrows show the direction of growth. (ex Lombardi & Wourms, 1988, modified). Further explanations see text. Right illustration (Fig. 17): Embryo of Ameca splendens at the peak of the development of the trophotaeniae.
6.4 The trophotaenia placenta
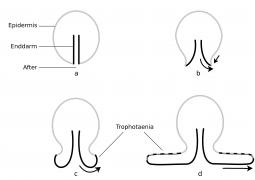
6.4.1 What is a trophotaenia placenta? Everyone seems to be familiar with what a placenta is; after all, there is a whole taxon (which includes humans) called "Placentalia" (= placental animals). It is often forgotten that there are numerous viviparous organisms that also have a placenta. The only question is how to define a placenta. One of the (numerous), very broadly applicable definitions states that a placenta already exists when maternal (or paternal) and embryonic tissue are so close to each other or even fused together that a physiological exchange between parent and child is ensured. It is tacitly assumed that this physiological exchange is about nutrients and not about respiratory gases or electrolytes (cf. Wourms et al., 1988, Greven, 1995, 2012). However, this definition also makes it clear that a placenta always consists of two parts, the embryonic and the maternal or the paternal (e.g. in seahorses). In goodeids, these parts are the trophotaenia of the embryos or the cell layer covering the trophotaenia and the cell layer lining the ovary cavity of the female (epithelium) with the underlying connective tissue.
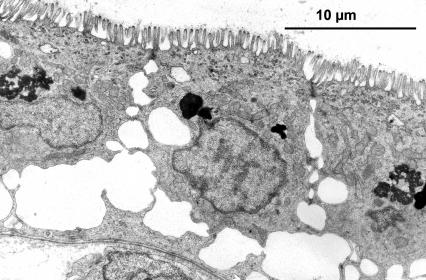
6.4.2 How does a trophotaenia placenta develop? The development of the trophotaenia probably only begins when the embryo is in the ovarian cavity. The earlier statements of Turner (e.g. 1933) and my earlier remark that the young remained in the follicle until the development of the trophotaenia (cf. Greven, 1995) is obviously not correct or does not apply to all goodeids in view of the findings described above, according to which very early stages of development were already found in the ovarian cavity. The trophotaenia can be formed very differently (Fig. 15). There are species that have four trophotaenia of different lengths (e.g. Girardinichthys spp.), others whose relatively short trophotaenia are arranged in a rosette around the anus (e.g. Goodea atripinnis), still others whose trophotaenia are shaped like irregular lobed bands (in Xenotaenia resolanae), have several partly branched bands (some Skiffia, Xenotoca and Ilyodon species, Ameca splendens), or consist of only two simple lateral bands (Characodon species) etc. These different formations are probably due to two main types, one band-shaped and one rosette-shaped (e.g. Lombardi & Wourms, 1988). However, the associated phylogenetic considerations (e.g. Turner, 1937, Hubbs & Turner, 1939) cannot be reconciled with molecular biological findings on the phylogeny of the goodeids. It must be assumed that the occurrence of the same type of trophotaenia in various apparently unrelated taxa, such as the number of four trophotaenia in Girardinichthys and Allotoca species, are homoplasies (homos, gr. = similar; plasis, gr. = formation), i.e. more or less phenotypically identical features that have arisen independently, i.e. convergently, in the different taxa. However, there is a trend within the goodeids to increase the surface area of the trophotaenia, e.g. by increasing the number of branches in trophotaenia of the ribbon type or by widening the individual trophotaenia in the rosette type (Doadrio & Domínguez-Domínguez, 2004). Ataeniobius toweri was long listed as the only goodeid species without trophotaenia. This is obviously not the case. Dawes (1995) has presented two pictures which seem to prove that embryonic stages of this species have accumulations of cells around the anus which resemble those of young embryonic stages of members of the genus Goodea. Although the published images are far from revealing important details (e.g. the surfaces of the cells, etc.), it must be assumed that this species has trophotaenia, but that these are extremely reduced and their occurrence is restricted to an early stage of development. This has been regarded as an basal characteristic, but considering more recent molecular biological findings on the position of A. toweri within the goodeids - A. toweri is then closely related to Goodea atripinnis (this was already suspected when looking at the karyotype) - a secondary regression of the trophotaenia must have taken place (Doadrio & Domínguez-Domínguez, 2004). The reduction of trophotaenia is thought to be correlated with a hypertrophied embryonic fin fringe, which connects the dorsal, anal and caudal fins (see Doadrio & Domínguez-Domínguez, 2004). Mendoza (1958) described such an enlarged fin margin in embryos of Goodea atripinnis and assumed that this extraordinarily richly vascularized structure was used for respiration, excretion and the absorption of nutrients from the ovarian fluid, especially as Goodea atripinnis did not exactly have conspicuously large trophotaenia. This has not yet been demonstrated experimentally. Vega-López et al. (2007) believe they have shown that the relatively large molecule vitellogenin is absorbed via the fin margin (of Girardinichthys). However, the findings presented so far are not convincing. It is certainly not wrong to assume a respiratory organ in the fin fold, which is richly supplied with blood. The first sign of the formation of trophotaenia, for example in Ameca splendens, is a thickening of the region around the anus (Lombardi & Wourms, 1988). This lengthens into a short tube, which splits as it continues to grow (Fig. 16a, b, c). These splits can reach considerable lengths in species with ribbon-shaped trophotaenia (Fig. 17). They are initially covered on the outside by the normal body epidermis and on the inside by typical resorbing (resórbere, lat. = to absorb) intestinal cells. Later, intestinal cells also migrate to the outside (Fig. 16d). Both cell types can be easily distinguished from each other (under a scanning electron microscope). The body cells have a pattern of micro ridges on their surface that resembles a fingerprint, the intestinal cells have small finger-shaped protrusions that are called microvilli (micrós, Greek for small; villus, Latin for villus) (Fig. 18). The epithelium of intestinal and somatic cells surrounds loose connective tissue in which some blood vessels are located (see Fig. 19). The maternal component of the trophotaenia placenta, the ovarian epithelium, and the connective tissue underneath it also change during pregnancy. Immediately under the epithelium there are numerous tiny blood vessels (capillaries; capíllus, lat. = hair), which increase considerably in number during pregnancy (Grossherr & Greven, unpublished). In addition, some species, e.g. Ameca splendens, show that the ovary epithelium itself is capable of producing protein-rich substances that are released into the ovarian lumen (Lombardi & Wourms, 1985), whereas this is not the case in other species such as "Xenotoca" eiseni and Xenoophorus captivus (e.g. Schindler, 1990, Schindler et al., 1988, Schindler & Kujat, 1990).
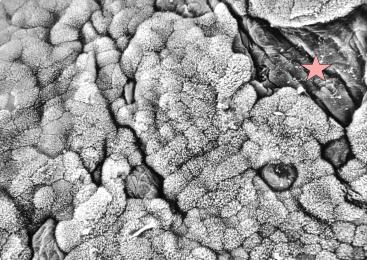
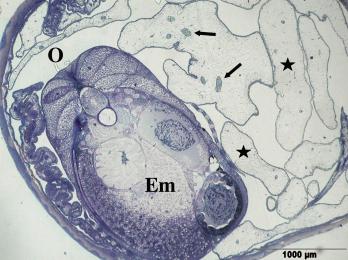
Left illustration (Fig. 18): Section of a trophotaenia of Neotoca bilineata with normal epithelium cells on the body surface (star) and gut cells (the rest). Scanning electron microscope picture (photo: G. Schindler). Right illustration (Fig. 19): Big embryo (Em) of Ameca splendens in the ovarium lumen (O). Please note the minor number of blood vessels (arrows) in the trophotaeniae (stars). Histological preparation.
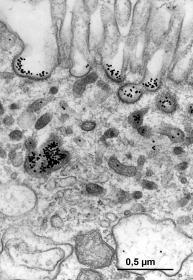
6.4.3 Trophotaenia placenta is not the same as trophotaenia placenta. Close contact between the maternal and embryonic components through thin epithelia, each consisting of a single cell layer, as well as an extremely rich blood supply, at least on the maternal side, strongly suggest an exchange of substances between mother and child. Nutrition and oxygenation of the young via such a trophotaenial placenta have been taken for granted since the discovery of these intestinal protrusions and have never been doubted. The importance of the trophotaenia can also be recognized by the fact that they enlarge or lengthen considerably in the course of embryonic development (see Fig. 17). They are only melted or fall off shortly before, and in some species even after, birth. In addition, numerous other functions have been attributed to the trophotaenia. For example, they are said to serve respiration (Turner, 1933, 1940; Mendoza, 1937) and to be involved in osmoregulation and excretion (Wourms et al., 1988). However, there is no clear evidence for any of these assumptions. However, the importance of the trophotaenia for the oxygen supply of the young has certainly been overestimated, as the supply of blood vessels in the nutrient cords is almost moderate compared to other regions of the body (Fig. 19). The body surface of the young, including the fins, appears to be better suited for this, as it is heavily supplied with blood (Schindler & Greven, 2010). For matrotrophy, i.e. the feeding of the growing young by the mother, the decisive factor is whether and to what extent substances can be absorbed by the trophotaenia that actually serve to feed the young. That this "must" be the case can already be concluded from the fact that trophotaenia at the peak of their development are for the most part also covered on the outer surface by intestinal cells (cf. also Wourms et al., 1988), which in general in "fish" (even in adults) are able to absorb large molecules via a process known as phago- (phagein, gr. = to eat) or more generally as endocytosis (éndon, gr, = inside; kýtos, gr. = cell). In addition, the trophotaenia of the growing young lie in a more or less viscous, mostly protein-rich fluid, so that it should not even be necessary and is probably not the case that all the trophotaenia of all the young lie close to the ovarian epithelium. This in turn would contradict the very "generous" definition of the placenta given above. Mendoza, who presented the first electron microscopic images of the trophotaenia of "Xenotoca" eiseni in 1972, postulated the uptake of molecules on the basis of the fine structure (typical absorbing intestinal cells; see Fig. 20), namely via the aforementioned endocytosis. However, this apparently does not mean that all possible molecules are taken up indiscriminately, but only very specific ones. Such selective endocytosis is referred to as receptor-controlled endocytosis. In this process, large molecules (e.g. certain proteins) from the ovarian fluid surrounding the young bind to special proteins (receptors; recípere, lat. = to absorb) in the membrane of the absorbing intestinal cell. The area of the cell membrane to which the proteins from the ovarian fluid are bound is invaginated into the interior of the cell. This invagination then constricts itself so that the molecules are enclosed in a vesicle inside the cell, where they can be put to further use, e.g. digestion within this vesicle. The uptake of large (protein) molecules can be visualized. Young goodeids removed prematurely from the ovary can be kept alive for a long time in physiological solutions and can be literally "fed". If, for example, such "nutrient" molecules are bound to tiny gold particles, which have the advantage that they are visible under the electron microscope because they scatter few electrons, their path can be followed from their attachment to the cell surface between the small projections of the intestinal cells, the so-called microvilli, right into the cell interior (Fig. 21). Trophotaenia are therefore ideal objects for studying the uptake and utilization of macromolecules. Since the first electron microscopic work by Mendoza (see above), their fine structure and ability to undergo endocytosis have been repeatedly studied morphologically in various species (e.g. Lombardi & Wourms, 1985, Grosse-Wichtrup & Greven, 1985, 1986, Schindler, 1990, Schindler & Kujat, 1990, Schindler & De Vries, 1986, 1987, 1988, Schindler & Greven, 1992, Kokalla & Wourms, 1994) and biochemically, so that we now also know more about the nature of the receptors involved (Schindler, 2003a). However, the fact that "Xenotoca" eiseni (Mendoza, 1972; Grosse-Wichtrup & Greven, 1985) and Ameca splendens (Lombardi & Wourms, 1985a, 1986, 1988) were initially used for such experiments was certainly a stroke of luck, because the trophotaenia of both species actually take up large molecules, are therefore capable of endocytosis and also possess the cellular equipment for this, which cannot be discussed in detail here. However, there are also species with trophotaenia that do not possess such an "endocytotic apparatus" for the uptake of large molecules, such as the cells of the trophotaenia of Girardinichthys viviparus (Schindler & De Vries, 1986, 1987) and the rosette-shaped trophotaenia of Goodea atripinnis (Hollenberg & Wourms, 1994). Surprisingly, however, the latter possess all the enzymes that characterize such an "endocytotic apparatus" (Kokkala et al., 1993). It is probably the case that the embryos of different goodeid species are not always supplied with comparable nutrients, but some are supplied with low molecular weight nutrients that do not have to be taken up via endocytosis, others with high molecular weight nutrients and still others with both types of nutrients. The molecules used as nutrients in the ovarian fluid ultimately come from the maternal circulation. There are proteins in the nutrient fluid that are obviously identical to proteins from the blood, but there are also proteins that are not identical to those in the blood and have therefore been modified or synthesized by the ovarian epithelium itself (cf. Hollenberg & Wourms, 1985, Schindler et al., 1988). In some species the ovarian epithelium also shows considerably increased secretory activity during pregnancy, G. atripinnis also has considerable fat reserves in the ovarian epithelium (Hollenberg & Wourms, 1985). The yolk proteins (vitellogenins) formed during embryonic development must also pass through the ovarian epithelium in order to be absorbed by the young. All in all, these findings also confirm what has already become clear from the dry weight studies described above, namely how different the degree of matrotrophy can be from species to species and that in some cases different molecules are also used to feed the young.
6.4.4 Adelphophagy: an additional or alternative food supply? Once the young have been freed from the egg membrane and the intestinal system has developed, it should also be possible to feed in the ovarian cavity in a "regular way", i.e. via the mouth. This could be the ovarian fluid, but certainly also its own siblings. Turner (1933) drew attention to the fact that a high percentage of embryos die in the ovarian cavity and are "absorbed" there (he assumed a regular digestion in the ovarian cavity, which of course is not conceivable). He also considered it likely that embryos or parts of them are absorbed by the siblings. This raises the question (which, incidentally, has not been clarified) of whether only dead siblings are eaten or whether living, weaker siblings are also eaten. In fact, oophagy (oón, gr. = egg; phagein, gr. = to eat) and adelphophagy (adelphós, gr. = brother), i.e. egg and sibling feeding, have been discussed for a number of goodeids and siblings as well as eggs have been documented, for example in the stomach of young Ameca splendens still in the ovary (Greven & Grossherr, 1992). According to what has been said above about development, however, the "eggs" could have been young embryonic stages. If this is the case, there must have been various developmental stages in the ovarian cavity, and this in turn would be an indication of a less successful superfetation. The dead or possibly still living siblings certainly represent an additional food source. Mendoza (1962) did not confirm that so many embryos actually die in the course of gestation - according to Turner (1933) it is about half to two thirds (!) - although, as he himself emphasizes, he did study other species. However, if this is confirmed in new studies, at least for some species, it would be interesting to find out whether this mortality rate, including adelphophagy, can be influenced. We have repeatedly observed in A. splendens and "Xenotoca" eiseni that individual females gave birth to very few, but particularly large young (see also Fig. 21). Perhaps adelphophagy is a mechanism for giving birth to a few young despite poor nutrition. If a female of a matrotrophic viviparous species does not have sufficient food during pregnancy, the supply of young would be jeopardized in the medium term, some of which could fall back on their weaker or dead siblings if the worst came to the worst. This is all pure speculation, but one could gather evidence for this by keeping a record of how many young are born under different nutritional conditions of the female, and whether these are conspicuously small or (if few are born) particularly large.
Left illustration (Fig. 20): Section of the resorbing trophotaenial epithelium of Ameca splendens. Please note the typical gut cells with finger-shaped evaginations (above, = microvilli) and the widened spaces between the cells. Transmission electron microscope picture. Right illustration (Fig. 21): Absorption of very big molecules (here: gold-particles coupled on proteins) through the resorbing cells of the trophotaeniae of Ameca splendens. Part of the gold particles are still outside of the cell and linked with the membrane of the microvilli, other particles are already in a vesicle inside of the cell. Transmission electron microscope picture.
6.4.5 Pregnancy and birth. Pregnancy begins with fertilization or karyogamy (= fusion of the nuclei of the egg and sperm cell; káryon, Greek = nucleus; gamein, Greek = mate). How and when this happens is unknown and eludes direct observation. Whether it can always be assumed as a first approximation that the mature oocytes are fertilized shortly after a successful copulation with a receptive female and the gestation period begins is questionable according to the findings of Mendoza (1937) on Neotoca bilineata. The latter drew attention to a special color change of the females of this species, which is apparently linked to the reproductive cycle. The females changed their abdominal coloration from light grey to vivid blue around the time of fertilization (about 10 days after the birth of the young). I have found nothing (more) about this phenomenon in the more recent literature. However, in order to obtain reasonably reliable data for a preliminary protocol that can be used to estimate the gestation period of goodeids, copulations that have been allowed in one day (by one male) can be considered as the time of onset of gestation (if the female becomes pregnant). To my knowledge, there is no immediate and visible sign (e.g. in behavior) in either the male or female goodeids that copulation has been successful, i.e. that spermatozoa have been transferred. Even though goodeids do not have a receptaculum seminis, i.e. they cannot store sperm over a longer period of time, the sperm will survive in the ovary for a short period of time. Turner (1937) even assumes that the females are also receptive during pregnancy, mate and therefore have sperm more or less continuously. This would also have to be proven first. As pregnancy progresses, the females become fuller and therefore certainly more immobile. Among other things, this must have a negative effect on the speed with which the females flee and their endurance when (fleeing) swimming etc.. Such a handicap would have to be accounted for on the cost side of the "viviparity" reproductive mode. In contrast to poeciliids, no further information is available for goodeids. The gestation period varies considerably between different species, but certainly also between females of the same species (e.g. depending on the temperature, the photoperiod, the availability of sufficient food, whether it is the first birth of the female or whether the female has given birth several times before, etc.), so that it makes little sense here to list data from the literature on gestation periods and intervals between individual births. The same applies to the number of pups born (although it is certainly worth keeping a record of this over a longer period of time under certain controlled husbandry conditions (cf. Mendoza, 1939, Arbuatti et al., 2012). Uribe et al. (2005) distinguish in a first approximation between species (tentatively extended by me on the basis of further literature data) that give birth to up to 60 young, such as Ameca splendens, Characodon lateralis, Goodea atripinnis, "Xenotoca" eiseni, species that give birth to up to 30 young, such as Neotoca bilineata, Alloophorus robustus, Girardinichthys viviparus, Xenotaenia resolanae, Zoogoneticus tequila, Xenoophorus captivus, Allotoca maculata, and species that only give birth to up to 15 young, such as Ataeniobius toweri, Chapalichthys encaustus and Skiffia francesae. However, the number can be greatly exceeded or undercut in individual cases. For example, Díaz-Pardo & Ortiz-Jiménez (1985) found an average of 27.5 embryos per female in Girardinichthys viviparus, but the maximum number in one female was 114. Young females giving birth for the first time and old females usually give birth to a smaller number of offspring (e.g. Mendoza, 1962, Kingston, 1978, Smith & Miller, 1980, Díaz-Pardo & Ortiz-Jiménez, 1985). The amount of offspring can apparently also be manipulated by varying the length of the day. Rueda-Jasso (2010) kept Skiffia lermae at different day lengths and obtained the most young at 16 h light and 6 h dark phase, but the heaviest and largest at 6 h light and 18 h dark phase.
The ovary of non-pregnant and pregnant Goodeids
Abstract
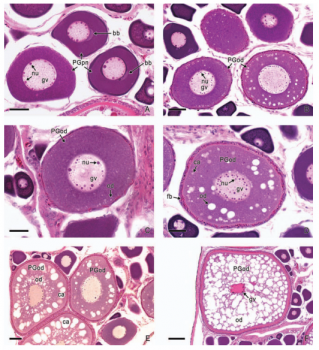
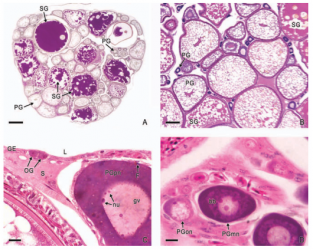
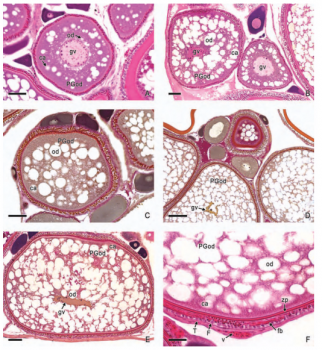
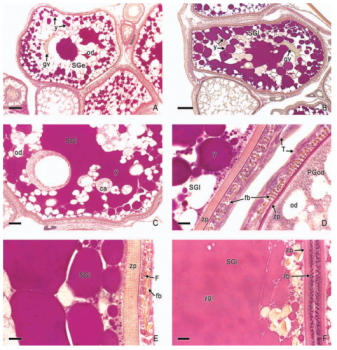
In viviparous teleosts, intraovarian gestation is the consequence of several reproductive characteristics: a) ovulation into the ovarian lumen, instead of into the coelom as result of the internal position of the germinal epithelium which lines the ovarian lumen, b) the development of internal fertilization, and c) the lack of oviducts. In viviparous teleosts, ovarian morphology is clearly modified, because teleost viviparity, invariably, involves the ovary in a gestational role. Therefore, the ovary of viviparous teleosts differs from those of all other vertebrates because it is the site, not only for production of eggs, but also for internal fertilization and gestation. This article illustrates reproductive structures in species of Goodeids, including the type of ovary during non-gestation, ovaries during early, middle and late intraluminal gestation, and trophotaeniae, extension of the embryonic gut to the lumen of the ovary, used for maternal-fetal metabolic exchange. The analysis of the ovarian morphology required for viviparity, and its similarities and differences comparing species, has special importance in the understanding of the events leading to this successful mode of reproduction. Based on both morphogenetic and physiological processes, the structure of the ovary in viviparous Goodeids possesses features that are unique among all vertebrates.
Introduction
The identification and definition of the female reproductive system, such as, the ovary, ducts and oogenesis, are fundamental elements in determining the reproductive pattern of a species. In particular, in the understanding of the teleost viviparity, the study of the ovary is essential to define the characteristics of the reproductive structures associated with this type of reproduction. The ovary of the species of the family Goodeidae presents special aspects that are unique in vertebrates. Some of them are similar to those belonging to the teleost fishes, others are similar to viviparous teleosts and, especially others are characteristics of goodeids. Therefore, it is convenient to analyze, initially, general features of 1) the ovary of teleosts and compare those with non-teleosts vertebrates, after that, distinguishes particularities of 2) the ovary of viviparous teleosts and, subsequently, identified the specific characteristics of 3) the ovary of Goodeids.
1. The ovary of teleosts
The ovary of teleosts is suspended to the dorsal wall by the mesovarium, between the swim bladder and the intestine. The embryological process of most teleost establishes the ovary differently from that of other vertebrates, becoming a sacular structure that contains a peripheral wall, and an internal lumen (Dodd & Sumpter, 1984). This type of process develops the ovary of teleosts in a sacular or cystovarian condition. The wall of the ovary forms irregular folds call lamellae that project into the ovarian lumen. A layer of epithelium, the germinal epithelium, borders the lamellae internally. This epithelium contains, among somatic cells, germinal cells. The germinal cells are the oogonia, the female stem cells that become eggs during their maturation. In contrast with this type of ovary, in non-teleost vertebrates, the germinal epithelium remains at the periphery of the ovary, in contact with the coelomic cavity (Dodd & Sumpter, 1984; Wake, 1985; Wourms et al., 1988; Grier et al., 2005; Uribe et al., 2005, 2009).
As a consequence of these two different types of ovarian developments in teleosts and non-teleost vertebrates, the ovulation occurs in different processes: a) in teleosts, at ovulation, the eggs are shed into the lumen of the ovary (intraluminal ovulation), or b) in non-teleost vertebrates, at ovulation, the eggs are shed into the coelomic cavity (external ovulation) (Wourms, 1981; Wake, 1985; Wourms et al., 1988; Guraya, 1989).
A complementary aspect of the cystovarian condition of teleosts is the way that the eggs follow from the lumen to the exterior of the body during the oviposition. This particular way defines another significant difference between teleosts and non-teleost vertebrates. Teleost do not develop oviducts, as occurs in the rest of vertebrates. Then, the caudal portion of the ovary of teleost, call gonoduct, communicates the ovary to the exterior of the body by the genital pore, the opening to the exterior. The gonoduct lacks germinal cells, consequently is formed exclusively by somatic tissues. This region is a peritoneal, muscular derivative of the caudal ovarian wall (Wourms, 1981; Dodd & Sumpter, 1984; Wake 1985; Wourms et al., 1988). In contrast, non-teleost vertebrates develop oviducts. The oviducts are the ducts that transport the oocytes from the coeloma to the exterior of the body during the oviposition; this is in the case of oviparous species or, in viviparous species, the embryos remain in a specialized region of the oviducts, the uterus, during gestation until the birth.
2. The ovary of viviparous teleosts
During early embryogenesis, in the majority of viviparous teleosts, the right and left ovaries fuse forming a single, sacular ovary. The ovary is of cystovarian type. The ovarian wall forms lamellae that extend into the ovarian lumen. The lamellae contain stroma with follicles in different stages of development (Wourms, 1981; Constanz, 1989; Lombardi, 1998; Uribe et al., 2005, 2009).
The number of eggs produced in viviparous teleosts during a gestational cycle is much lower than occurs in oviparous teleosts (Grier et al., 2005). Meanwhile in oviparous species thousands or hundreds of thousands of egg mature during the reproductive cycle, in viviparous species dozens of eggs or less mature in the reproductive cycle. Thibault and Schultz (1978) revise the two extremes of having high reproductive rates and low survival rates, in oviparous species; or low reproductive rates and high survival rates, in viviparous species.
In the evolution of viviparity the insemination and internal fertilization are fundamental pre-requisites. Insemination is the transfer of sperm from the male to the female gonoduct that permit the internal fertilization; that is, the fusion of sperm and egg within the ovary (Greven, 2005). It is necessary that the spermatozoa be able to live before fertilization, at least, for short periods of time, in the fluid secreted by the germinal epithelium contained in the ovarian lumen. The presence of spermatozoa inside the ovary of viviparous teleosts has revealed another special characteristic of the germinal epithelium, also, unique among vertebrates (Turner, 1947).
As a consequence of the cystovarian condition and the lack of oviducts in teleosts, the gestation in viviparous species occurs inside the ovary, emerging an intraovarian gestation. That means, the embryos are developed inside the ovary until birth. The intraovarian gestation is characteristic of viviparous teleosts and it is unique among vertebrates. The intraovarian gestation is the result of the confluence of three main reproductive aspects in viviparous teleosts, already mentioned: a) ovulation into the ovarian lumen instead of into the coelom; b) insemination and internal fertilization, and c) absence of oviducts (Wake, 1985; Wourms et al., 1988).
There are two different processes for providing embryos with nourishment during gestation in viviparous teleosts, both involving ovarian components and egg morphology. These processes are: lecithotrophy and matrotrophy (see review Wourms, 2005). In lecithotrophy, the energy substrates are contained in abundant yolk reserves that are stored in the egg during oogenesis, prior to fertilization. In matrotrophy, the energy substrates are provided, not only by those stored in the egg, but also by supplies from the maternal blood vessels of the ovarian tissues during the course of embryonic development, after fertilization (Turner, 1947; Amoroso, 1981; Wourms et al., 1988; Wourms & Lombardi, 1992; Lombardi, 1998). Wourms (2005) suggested that the trophic relationships in the evolution of viviparity involve the transition from lecithotrophy to matrotrophy, a shift from embryonic nutrition from the stored reserves in the yolk, to maternal nutritional dependency. Consequently, the reduction of yolk of the egg involves matrotrophy (Turner, 1940; Wourms et al., 1988). Wourms (2005) commented that the most advanced type of viviparity occurs in species in which the yolk has become greatly reduced, and the embryos develop adaptations for exchanging nutrients from the maternal blood, as occurs in Goodeids. Goodeids are remarkably different from other viviparous fishes with regarding to reduction of egg yolk.
Considering all of these morphogenetic processes observed in the ovaries of viviparous teleosts, the structure of this type of ovary, and its adaptations for viviparity are relevant among vertebrates. Thus, the analysis of the ovarian morphology required for viviparity, and its similarities and differences comparing species, as goodeids, has special importance in the understanding of the events leading to this successful mode of reproduction.
3. The ovary of Goodeids
The family Goodeidae includes two subfamilies: the oviparous Empetrichthyinae, comprising the two Nevada genera (Empetrichthys and Crenichthys) and a fossil species from California and the viviparous Goodeinae of the Mexican Plateau (Parenti, 1981).
As observed in other viviparous teleosts, the ovary of viviparous Goodeids is a single, saccular, elongated structure (Fig. 1A), that is suspended dorsally by peritoneal mesentery, the mesovarium. A peripheral wall and a central lumen form the ovary (Figs. 1A,B, 2A-D). The caudal region of the ovary forms the gonoduct (Fig. 1C). The gonoduct contains thick muscle layers. The limit between the gonoduct and the germinal region of the ovary has large and thin folds (Uribe et al., 2010). Particular features of the ovary of viviparous Goodeids are described in several species of goodeids (Turner, 1933; Hubbs & Turner, 1939; Mendoza, 1939, 1941, 1943; Schindler, 1990; Uribe et al., 2004), and are summarized by Uribe et al. (2005) in the following aspects:
The lumen is partially or completely divided by a septum (Figs. 1A, 2A-C). According to the species, the septum could be unfolded and thin as in the genera: Ilyodon, Skiffia (Figs. 2A,B ) and Girardinichthys, or folded and thick as in the genera: Goodea and Xenotoca (Fig. 2C). In some species, the septum is incomplete, forming two different flaps as in the genera: Ataeniobius and Neoophorus; or forming only one flap as in the genus: Xenoophorus (Hubbs & Turner, 1939; Uribe et al., 2005, 2009).
Left illustration: Sagittal sections of the ovary of Goodea atripinnis (A, B) and the gonoduct of Skiffia multipunctata (C). A, B: during non gestation (A), gestation (B). The ovigerous tissue (W) forms irregular folds and lamellae (LA) with follicles (F) in different stages of maturation; even the wrinkled septum (S) contains follicles. The stroma (st) is comprised mainly out of loose vascularized connective tissue. Ovarian lumen (L). C: Gonoduct (G); the tissue (W) of the gonoduct (G) posseses a layer of smooth muscles (M); its folds project into the lumen (L). Scale: A = 100 µm. B = 50 µm. C = 50 µm. Right illustration: Transverse sections through the ovaries of non gestating Ilyodon whitei (A, B) and „Xenotoca“ eiseni (C, D). The ovigerous tissue (W) ist confined externally by the serosa (Se). The lamellae contain follicles (F), that are surrounded by stroma (st) und covered by the germinal epithelium (Ge). Lumen (L). A, B: The ovary of this species forms a lateral fold or lamella (La) on each side; the septum (S) is not wrinkled; its epithelium contains no germ cells. C, D: This type of ovary has various, irregular formed lamellae, the septum is heavily wrinkled. Lamellae and septum contain germ cells. Scale: A = 100 µm. B = 50 µm. C = 100 µm. D = 30 µm
The wall of the ovary forms irregular lamellae that project into the lumen (Figs. 1A, 2A-D). The lamellae are lined by the germinal epithelium (Figs. 2B,D) and include vascularized loose connective tissue and follicles in different stages of development (Figs. 1A,B, 2A-D) (Uribe et al., 2009). The oogonia, contained in the germinal epithelium, proliferate by mitosis (Fig. 3A), initiate meiosis becoming oocytes and enter in oogenesis. Gradually, follicular cells surround the oocytes developing the follicles, where oocytes mature. The ovarian histology of several Goodeids has been described, as in Neotoca bilineata (Turner, 1933; Mendoza, 1941, 1943; Dodd & Sumpter, 1984), Ilyodon whitei and Goodea atripinnis (Uribe et al., 2004, 2005, 2009). Histologically, the ovarian wall of Goodeids consists of four tissue layers. From the interior to the exterior they are: a) germinal epithelium; b) stroma, formed by loose vascularized connective tissue and follicles in different stages of development; c) smooth muscle layers; and d) serosa, formed by scarce connective tissue and mesothelium (Figs. 2A-D).
The germinal cells could be dispersed throughout the germinal epithelium and the stroma all around the ovarian wall and also in the septum, as in the genera: Allotoca, Ataeniobius, Goodea, Neoophorus and Xenotoca (Fig. 2C), or they could be concentrated in special regions of the ovarian wall and the septum lack germinal cells, as in the genera: Girardinichthys, Ilyodon (Fig. 2A) and Skiffia (Hubbs & Turner, 1939).
The oogenesis of Goodeids presents four stages according to the classification for oogenesis of teleost defined by Grier et al. (2009): a) Oogonia Proliferation Stage (OP): the oogonia divide mitotically in the germinal epithelium (Fig. 3A); b) Chromatin Nucleolus Stage (CN): upon the initiation of meiosis, the oogonia enter in oogenesis and they develop into early oocytes, when they become progressively surrounded by a single layer prefollicle cells and form primary follicles. During this stage the oocytes are located in groups, called cell nests (Figs. 3A,B); c) Primary Growth Stage (PG): the growth of the follicles includes previtellogenesis with increase of ooplasm (Figs. 3A-C) and later incorporate lipid droplets (Fig. 3D); and d) Secondary Growth Stage(SG): when vitellogenesis is going on with gradual deposition of yolk (Figs. 4A-C). Progressively, in the mature oocyte, the yolk becomes fluid and the nucleus moves to the animal pole of the oocyte (Fig. 4D).
Left illustration: Ovarian follicles of „Xenotoca“ eiseni (A), Ilyodon whitei (B), Girardinichthys multiradiatus (C) and Skiffia multipunctata (D) during the proliferation (o) of the oogonia, during the chromatin-nucleolus-phase (CN) and in phase of primary growth (PG). A, B, C: In the lamellae are located partially in nests arranged oogonias in proliferation (o) - please note the dividing cell with metaphase chromosomes - and oocytes in CN-phase. In the lamellae are also located oocytes in PG-phase (basophilic cytoplasm, big and globular nucleoli (n)). The oocytes are surrounded by a layer of follicle cells (fc). D: Follicle during PG-phase with numerous drops of oil (od) within the cytoplasm of the oocyte and an irregular shaped nucleolus (n). Follicle cells (fc), germ epithelium (Ge), ovarian lumen (L), stroma (st). Scale: A = 15 µm. B = 30 µm. C = 30 µm. D = 30 µm Middle illustration: Ovaries of Goodea atripinnis (A), „Xenotoca“ eiseni (B, C) und Skiffia multipunctata (D) with follicles in phase of secundary growth (SG). A: Lamellae of the ovarian tissue (W) with follicles during SG-phase and PG-phase. Start of the vitellogenesis (y). B, C: Lamellae of the ovarian tissue (W) with oocytes during SG-phase with liquidizing yolk (fy), and during PG-phase. Nucleolus (n) in centre of oocyte, surrounded by a layer of follicle cells (fc). D: Follicle during SG-phase with liquified yolk (fy) and nucleolus (n) being translocated to the animal pole, and follicle in PG-phase. Spermatozoa (z) in lumen (L) of the ovary. Germ epitehlium (Ge), stroma (st). Scale: A =100 µm. B = 50 µm. C = 100 µm. D =150 µm. Right illustration: Ovaries of Ilyodon whitei (A) und Girardinichthys multiradiatus (B). A: Indentation (dl), that extends with a thin duct from the ovarian lumen (L) near a follicle in PG-phase. The oocyte contains numerous drops of oil (od) and a centrally located nucleolus (n). Germ epithelium (Ge). B: Numerous spermatozoa (z) in the ovarian lumen (L) and on the germ epitehlium (Ge). Stroma (st). Scale: A = 50 µm. B = 10 µm.
The mature oocytes of Goodea atripinnis attains 0.8-1.3mm in diameter. Meanwhile, the mature oocytes of Ilyodon whitei, Girardinichthys viviparus and Characodon lateralis attain 0.8-1mm in diameter (Turner, 1940; Mendoza, 1972; Uribe et al., 2005). During gestation there is not development of oocytes. Only after birth, the oogenesis become activated and develops maturation of new oocytes, and after insemination and fertilization a new embryonic development is initiated.
In Goodeids, spermatozoa are seen in the ovary only during the time around the fertilization (Uribe et al., 2005), when the oocytes attain maturation until early embryonic development (Figs. 4D). Fine tracts branching from the ovarian lumen to the oocytes communicate the lumen with the follicles. These tracts are called delle by Stühlmann (1887) (Fig. 5A). During fertilization, the spermatozoa enter through de delle and reach the follicle. The spermatozoa are seen dispersed in the ovarian lumen (Fig. 4D) and close to the germinal epithelium (Fig. 5B). Fertilization occurs when the mature oocyte is inside the follicle, in an intrafollicular fertilization, consequently, in viviparous goodeids there is not ovulation (Turner, 1947; Wourms et al., 1988; Uribe et al., 2005). After intrafollicular fertilization, embryonic development is initiated within the ovarian follicle. Then, the embryos are retained in the follicle for a brief period of time, and during early embryogenesis (blastodisc or neurula), the embryos are discharged from the follicle into the ovarian lumen (Fig. 6A), process call embryonation (Uribe et al., 2010), where development continues until birth. This type of gestation is identified as intraluminal gestation (Turner, 1940, 1947; Amoroso, 1960, 1981; Wourms, 1981; Wourms et al., 1988; Schindler et al., 1988: Greven et al., 1993). Embryos developing in the ovarian lumen of Ilyodon whitei during blastodisc stage (Fig. 6A), Skiffia multipunctata during neurula (Fig. 6B), Ilyodon whitei during middle development (Fig. 6C), and late development (Fig. 6D), (Fig. 6A), evidence the intraluminal gestation.
Left illustration: Ovaries of Ilyodon whitei (A, C, D) and Skiffia multipunctata (B) during the intraluminal gestation. A: Sagittal section with early embryo (Discoblastula, eE) in the ovarian lumen. The embryo still contains plenty of yolk (fy) below the discoblastula. B: Transversal section with early embryo (Neurula, eE). C: Sagittal section (medially gestation stage) with several further developed embryos (mE) and beginning of the gonoduct (G). Ovigerous tissue (W). D: Sagittal section; embryos in a late gestation stage (lE). Follicle (F), gonoduct (G), lamellae (La), ovarian lumen (L), ovigerous tissue (W), septum (S). Scale: A = 300µm. B =120 µm. C = 2 mm. D = 2 mm. Right illustration: Trophotaeniae of Ilyodon whitei (A) and „Xenotoca“ eiseni (B, C). A: Sagittal section of an embryo (lE, late gestation) with ribbon type trophotaeniae (T), in ovarian lumen. Gut (g), liver (l), nervous system (ns) (ex Uribe et al. 2005). B, C: Embryo, late gestation (lE) with mit trophotaeniae (T). Gut (g), muscles (m), nervous system (ns), ovarian lumen (L), septum (S). Scale: A= 1,5 mm. B = 50 µm. C = 30 µm
The germinal epithelium in Goodeids might acquire several and complex functions, becoming modified in the support of the intraluminal gestation. The germinal epithelium is involved in the secretions of specific substances and exchange of multiple metabolites from the maternal vascular system, located in the stroma just beneath the germinal epithelium, to the ovarian fluid. This complex fluid contained into the lumen is the histotrophe that bathes the developing embryos and provides nutrition (Turner, 1940, 1947; Schindler et al., 1988; Schindler & Hamlett, 1993). Mendoza (1939, 1941) described the cyclic changes of the ovarian structure of Neotoca bilineata during non-gestation and gestation stages, showing an increase in the number of secretory cells in the germinal epithelium during gestation, and the elaboration of vast quantities of secretion. Schindler et al. (1988) observed in Xenoophorus captivus the increase in cell size and secretory activity of the germinal epithelium during gestation. The germinal epithelium is also involved in the removal of the waste products of the embryonic metabolism. Schindler et al. (1988), and Wourms et al. (1988), assumed that waste products are, evidently, transported across the ovarian germinal epithelium and are subsequently removed via the maternal vascular system.
In Goodeids, the quantity of yolk contained in the egg is too small to supply the embryos with all the necessary nutrients throughout gestation. In fact, soon after the embryos are released into the ovarian lumen, their small supply of yolk is absorbed by the embryo, during middle gestation. Then, lecithotrophy is finished. Later, the hindgut develops extensions, the trophotaeniae (Figs. 7A-C), that project into the ovarian lumen (Turner, 1937; Wourms, 1981, 2005; Wourms et al., 1988; Schindler & Hamlett, 1993; Greven et al., 1993). The trophotaeniae absorbs nutrients derived from the histotrophe, then, matrotrophy is initiated. Lombardi and Wourms (1988) indicated that the development of the trophotaeniae manifests the shift from the primitive lecithotrophy to a more specialized state, the matrotrophy.
Trophotaeniae are characteristic in all species of viviparous Goodeids; even in Ataeniobius toweri that has rudimentary trophotaeniae, described as short, hemispheric structures of external gut tissue surrounding the vent (Wourms, 2005). As embryonic development proceeds, Goodeid embryos develop progressively the trophotaeniae, and increase the absorption of maternally-supplied nutrients. Wourms (2005) examined the function, morphology, development, and evolution of trophotaeniae.
The trophotaeniae are composed of vascularized loose connective tissue, surrounded by simple columnar epithelium, similar to that of the gut absorptive epithelium. The trophotaenial epithelium has a great absorptive efficiency for taking up nutrients contained in the histotrophe. Several authors observed the trophotaenial epithelial cells of Goodeids, as in "Xenotoca" eiseni (Mendoza, 1972), Xenoophorus captivus (Schindler & de Vries, 1987), Ameca splendens, Goodea atripinnis (Lombardi & Wourms, 1988; Kokkala & Wourms, 1994), "Xenotoca" eiseni (Greven et al., 1993), Allophorus robustus, Ilyodon furcidens, Zoogoneticus quitzeoensis (Hollenberg & Wourms, 1994), describing an apical brush-border in the epithelial columnar cells, a morphological feature that indicates absorptive activity. Trophotaenial ecdysis occurs at parturition when undergo degeneration and are lost (Turner, 1937; 1972; Wourms et al., 1988).
There are two types of trophotaeniae in Goodeids (see reviews by Lombardi, 1998; Wourms, 2005): a) rosette type, which consists of short, lobulated, folded processes, as in the genera Allotoca, Goodea, Neoophorus, Xenoophorus; and 2) ribbon type, which consists of long, thin, flattened processes, as in the genera Allophorus, Ameca, Chapalichthys, Characodon, Girardinichthys, Ilyodon (Fig. 7A), Skiffia (Figs. 7B,C), Xenotoca, Zoogoneticus. The rosette and ribbon types of trophotaeniae indicate different levels of nutrient transfer efficiency. In the ribbon type there is an amplified surface area that is produced by the elongation, as compared to the rosette type. Lombardi and Wourms (1988) mention that the increased of nutrient transfer efficiency is associated with increased of absorptive surface area as in the ribbon type trophotaeniae (Wourms & Lombardi, 1992; Hollenberg & Wourms; 1994; Schindler & de Vries, 1987; Schindler, 1990; Greven et al., 1993). Therefore, ribbon trophotaeniae are considered to be the most advanced type (Wourms, 2005). Mendoza (1972) has suggested that the trophotaeniae are a fundamental innovation that was developed by goodeids in their evolutionary adaptations to the exigencies of viviparity and represent a culmination in the evolutionary sequence of adaptations of teleosts in the light of gut ontogeny (Wourms & Lombardi, 1992).
In the natural habitat, gestation requires approximately 6 to 8 weeks and reproduction occurs continuously during summer months. Comparing the number of developing embryos in several species of Goodeids, there are differences in fecundity. There are three groups of Goodeidae according to data on fecundity: a) Species with 50-60 embryos as in Ameca splendens, Characodon lateralis, Goodea atripinnis, "Xenotoca" eiseni; b) Species with 20-30 embryos as in Neotoca bilineata, Allophorus robustus, Girardinichthys viviparus, Xenotaenia resolanae, Zoogoneticus tequila, Xenoophorus captivus; c) Species with 5-15 embryos as in Ataeniobius toweri, Chapalichthys encaustus (Mendoza, 1962; Meyer et al., 1985; Webb & Miller, 1998; Uribe et al., 2005). However, females in their first year of reproduction may give birth fewer than 10 young (Webb & Miller, 1998) and similar number of young in females in advanced age (Grier & Uribe, unpublished). Additionally, the fecundity could be dependant of the female size. The embryos occur in both chambers of the ovarian lumen. Prior to birth, the young Goodeids are nearly 1.3-1.8cm in length. The position of the young into the ovary is longitudinal, parallel to the major axis of the ovary.
Conclusions
Intraovarian gestation in the ovary of Goodeids is possible because the confluence of several morphological characteristics: a) the ovary of cystovarian type; b) the germinal epithelium lining the ovarian lumen; c) the lack of oviducts; and, d) the internal fertilization. Additionally, the intraluminal gestation is a consequence of: e) the discharge of embryos into the lumen after intrafollicular fertilization; f) the secretion of histotrophe to the lumen; and, g) the development of trophotaeniae. After fertilization, Goodeid embryos are retained in the follicle for only a brief period of time. During early embryogenesis, they are discharged from the follicle into the ovarian lumen where the development continues. In Goodeids, the quantity of yolk contained in the egg is too small to supply the embryos with all of the nutrients that are necessary throughout gestation. Trophotaeniae are characteristic in all species of Goodeids. The development of trophotaeniae in Goodeids represents a culmination in the evolutionary sequence of adaptations of teleosts in the light of gut ontogeny (Wourms, 2005). Based on both morphogenetic and physiological processes, the structure of the ovary in viviparous Goodeids possesses features that are unique among all vertebrates.
Ovarian structure and Oogenesis of the Oviparous Goodeids Crenichthys baileyi (Gilbert, 1893) and Empetrichthys latos, Miller 1948 (Teleostei, Cyprinoontiformes)
ABSTRACT
The cyprinodontiform family Goodeidae comprises two biogeographically disjunct subfamilies: the viviparous Goodeinae endemic to the Mexican Plateau, and the oviparous Empetrichthyinae, known only from relict taxa in Nevada and California. Ovarian characteristics of two oviparous species of goodeid, Crenichthys baileyi and Empetrichthys latos, studied using museum collections, are compared with those of viviparous species of goodeids. Both subfamilies have a single, cystovarian ovary. The ovary in the viviparous Goodeinae has an internal septum that divides the ovarian lumen into two compartments, and it may possess oogonia. There is no ovarian septum in the oviparous C. baileyi and E. latos. Oogenesis is similar in both subfamilies with regard to the proliferation of oogonia, initiation of meiosis, primary growth and development of an oocyte during secondary growth in which fluid yolk progressively fuses into a single globule. Notably, eggs of C. baieyi and E. latos are approximately double the size of those of the viviparous Goodeinae in which embryos develop inside the ovarian lumen and are nourished, in part, by nutrients transferred from the maternal tissues, a mode of embryo development called matrotrophy. Egg envelopes of the two subfamilies differ in that those of C. baileyi and E. latos have a relatively thick zona pellucida, attachment fibrils or filaments that develop between the follicle cells during oogenesis, and a micropyle observed only in E. latos. In contrast, viviparous goodeid eggs have a relatively thin zona pellucida, but lack adhesive fibrils, and a micropyle was not observed. These reproductive characters are compared with those of species of the eastern North American Fundulus, a representative oviparous cyprinodontiform. One newly recognized shared, derived character, a single, median ovoid ovary with no obvious external evidence of fusion, supports monophyly of the Goodeidae. Differences among the goodeid subfamilies and Fundulus are interpreted relative to the oviparous versus viviparous modes of reproduction.
INTRODUCTION
The viviparous goodeid fishes have long been celebrated in fish reproductive biology for their unique trophotaeniae: short or elongate, somewhat elaborate extensions from the hindgut in embryos and neonates that function in nutrition and respiration (e.g., Turner, 1933, 1937, 1940; Mendoza, 1937, 1940, 1943, 1965; Hubbs & Turner, 1939; Wourms, 1981, 2005; Wourms et al., 1988). Ovarian structure and oogenesis have been described in comparable detail (Turner, 1933, 1947; Mendoza, 1940, 1943; Wourms, 1981, 2005; Wourms et al., 1988; Schindler & Hamlett, 1993; Uribe et al., 2004, 2005, 2009). Anatomical variation in the ovary and the trophotaeniae formed the basis of an early systematic classification (Hubbs & Turner, 1939). Today, the viviparous goodeids comprise 17 genera with 41 species, all endemic to the Altiplano of Central Mexico (Domínguez-Domínguez et al., 2005). Their unique form of viviparity and extensive diversification in freshwater habitats restricted to the Mexican Plateau has generated interest in conservation of the native freshwater fish biota of Mexico: two, possibly three, species are extinct and nearly all others are under some risk (Domıínguez-Domínguez et al., 2005).
The relationships of viviparous goodeids to other cyprinodontiforms were unspecified until Parenti (1981) hypothesized that the closest living relatives of the viviparous goodeids are two relictual, oviparous freshwater killifish genera that live in southern Nevada, Crenichthys and Empetrichthys. The family Goodeidae, therefore, was expanded to comprise two subfamilies: the viviparous Goodeinae of the Mexican Plateau and the oviparous Empetrichthyinae, comprising the two Nevada genera and a fossil species from California. Both groups share derived characters of the oral jaws and the anal fin (Parenti, 1981). The first 6 or 7 anal fin rays of male viviparous goodeids are shortened and unbranched and separated from the rest of the fin. In all goodeids, oviparous and viviparous, the first 2 to 7 middle anal radials are fused to the proximal radials. The sister-group relationship of the Goodeinae and Empetricthyinae was corroborated in subsequent morphological (e.g., Costa, 1998) and molecular (Grant & Riddle, 1995; Webb et al., 2004) analyses. The Empetrichthyinae comprises 10 recent species or subspecies: Crenichthys baileyi, with five subspecies C. b. baileyi, C. b. albivallis, C. b. grandis, C. b. moapae, C. b. thermophilus, and C. nevadae; Empetrichthys merriami and Empetrichthys latos, with three subspecies E. l. latos, E. l. pahrump, and E. l. concavus (Soltz & Naiman, 1978; Williams & Wilde, 1981). They lack pelvic fins and share a unique form of the first epibranchial (Parenti, 1981: Fig. 47b; Fig. 1). A third species, Empetrichthys erdsi, is known only as a fossil from the Los Angeles basin, California (Uyeno & Miller, 1963). They were once somewhat more broadly distributed throughout springs, pools, and streams of the Great Basin and Mojave deserts of Nevada and southern California (Gilbert, 1893) and, like their Mexican sister-group, today suffer intense habitat deterioration and degradation (Minckley & Marsh, 2009). E. merriami and all but one subspecies of E. latos are extinct (Minckley & Deacon, 1968; Miller et al., 1989). The extant E. l. latos, endemic to a single spring in Manse Ranch from which it has been extirpated, persists in refugia outside ist native Pahrump Valley (Soltz & Naiman, 1978; Minckley & Marsh, 2009). C. baileyi is listed as threatened (Minckley & Marsh, 2009). In contrast to our extensive knowledge of viviparous goodeid reproduction, we are aware of only the briefest details of reproduction of empetrichthyines (Hubbs, 1932; Hubbs & Miller, 1948; Miller, 1948; Kopec, 1949). Wild-caught C. baileyi laid 10–17 eggs, one at a time, during spawning events. Eggs measured 1.9 mm in diameter, and eggs had adhesive fibrils or filaments by which they are attached to vegetation (Kopec, 1949). Fertilized eggs hatched in 5–7 days in the laboratory (Kopec, 1949). Reproduction is asynchronous; females spawn at least twice per year (Minckley & Marsh, 2009). There are no studies of Empetrichthys or Crenichthys ovarian morphology and oogenesis. Taxa of both genera are extinct, endangered, or threatened. We were fortunate to have the opportunity to examine archival collections made in the mid 1960s and stored at Arizona State University (ASU). In addition to comparing the ovaries and oogenesis in both oviparous and viviparous goodeids, we include data on the oviparous cyprinodontiform, Fundulus, for outgroup comparison. Here, we describe the morphology of the ovary and the process of oogenesis of C. baileyi and E. latos.
RESULTS
Histological elements of the ovary are germ cells (oogonia and oocytes in different stages of development) and somatic tissues (somatic cells of the germinal epithelium, vascularized stroma, smooth muscle, and peritoneum). Even though the ovaries processed in this study were fixed in the mid 1960s, the histological preparations revealed good preservation of ovarian tissues. The histological characters of the ovaries and of oogenesis of C. baileyi and E. latos are comparable; our descriptions of the ovary and of oocyte development pertain to both species. The illustrations of C. baileyi histology are presented in Figures 2–5, and that of E. latos in Figures 6–9; images of both species are combined in Figure 10.
Ovarian Components
C. baileyi and E. latos have a single ovary, ovoid in shape, and longitudinally suspended from the dorsal wall by the mesovarium. The ovary is of cystovarian type, containing a central lumen. Irregular lamellae or folds project from the ovarian wall into the lumen. Lamellae are composed of stroma that exhibits a diversity of follicular stages of development (Figs. 2A,B and 6A,B). The surface of the lamellae is lined by the germinal epithelium. The germinal epithelium, as are all epithelia, is separated from the stroma by a basement membrane. The germinal epithelium contains oogonia and early oocytes that are scattered among somatic epithelial cells (Figs. 2C and 6C) or as extensions of the germinal epithelium that form cell nests. Each follicle is encompassed by a basement membrane which separates it from the stroma.
Oogenesis and Folliculogenesis
Oogenesis and folliculogenesis in both C. baileyi and E. latos begins when oogonia proliferate and enter meiosis to become oocytes. During folliculogenesis, early oocytes in cell nests are enclosed progressively by somatic epithelial cells that become prefollicle cells. The prefollicle cells, in turn, become follicle cells when they, and the oocyte, form a follicle (Figs. 2D,E and 6C). They are then completely surrounded by a basement membrane. Subsequently, a thin, vascularized theca, derived from the stroma, develops around the basement membrane. The structure formed by the follicle (oocyte and follicle cells), and surrounding elements (basement membrane and theca) form the ovarian follicle complex. Subsequently, oocytes undergo morphological and physiological changes throughout the stages of oogenesis defined as in the classification of Grier et al. (2009).
Oogonia Proliferation Stage
Oogonia are scattered among somatic epithelial cells in the germinal epithelium (Figs. 2C–E and 6C). Oogonia are spherical cells with an approximate diameter of 8–10 μm. The ooplasm is hyaline. The nucleus is spherical and contains a single nucleolus and fine granular chromatin. The oogonia initiate meiosis, entering the CN stage of development; then they are oocytes.
Chromatin-Nucleolus Stage
The oocyte is in CN stage (Fig. 2D,E) when meiosis begins. It advances through leptotene, zygotene, pachytene, and early diplotene of prophase I of meiosis when, gradually, the chromosomes condense and are seen as thin threads. During early diplotene, lampbrush chromosomes develop and the oocyte begins to grow, reaching 10–12 μm in diameter. The ooplasm of early diplotene oocytes is hyaline, similar to that seen in the oogonia, as folliculogenesis proceeds. The prefollicle cells completely enclose the oocyte, beginning to define the developing follicular structure.
Primary Growth Stage or Previtellogenesis
The PG stage comprises: one nucleolus step (PGon), multiple nucleoli step (PGmn), perinucleolar step (PGpn), and oil droplets—cortical alveoli step (PGod). One nucleolus step (PGon). The size of the oocytes increases to 40 μm. The nucleus of the oocyte, now called a germinal vesicle, is spherical and enlarges in volume, reaching 15 μm in diameter. The germinal vesicle contains chromosomes that are arrested in the diplotene step as characteristic lampbrush chromosomes, and a single, spherical nucleolus (Figs. 2D and 6D). The ooplasm becomes progressively more basophilic as ribonucleic acids are synthesized in the germinal vesicle by the nucleolus and are transported to the ooplasm. The follicle cells form a squamous cell layer (2–3 μm high), completely surrounding the oocyte. Multiple nucleoli step (PGmn). The oocytes attain a diameter of 80 μm. The oocyte’s germinal vesicle now contains multiple nucleoli (Figs. 2D–F and 6D) that are randomly distributed. Basophilia of the ooplasm is intense, as in the previous step. The Balbiani bodies, irregular structures of the ooplasm adjacent to the germinal vesicle, are seen (Figs. 2E,F and 6D). The follicle cells form a squamous cell layer, similar to those in the previous step. The basement membrane and the vascularized theca completely surround the follicle. Perinucleolar step (PGpn). The oocyte attains a diameter of 150 μm. The germinal vesicle is spherical and large, about 60 μm in diameter. The multiple nucleoli become oriented around the inner membrane of the germinal vesicle (Figs. 3A and 6C). The nucleoli are spherical and have varying diameters (2–4 μm). Balbiani bodies are around the periphery of the germinal vesicle (Fig. 3A). There is a slight decrease in ooplasmic basophilia. The follicle cells remain squamous, as in the preceding two steps. The vascularized theca is similar to those in the previous step. Oil droplets and cortical alveoli step (PGod). The oocytes reach 550 μm in diameter. Multiple nucleoli are positioned around the periphery of the germinal vesicle. Oil droplets and cortical alveoli appear around the germinal vesicle, later, they are dispersed throughout the ooplasm. Spherical oil droplets (Figs. 3B–E and 7A–C) increase progressively in number and size; some of them attain 50 μm in diameter. Cortical alveoli are spherical and vary in diameter (Figs. 3D,E and 7A–C). At first appearance, some of them are 2 μm in diameter, but later they may reach 25 μm. Then, there is a massive accumulation of oil droplets throughout the ooplasm (Figs. 3F and 7D,E). The zona pellucida is well defined (Figs. 4A,B and 7F) as a homogeneous line, densely stained, 2–3 μm thick. At the end of this step, the zona pellucida (Figs. 4B and 7F) reaches 10–12 μm thick and is clearly striated. The follicle cell layer (Fig. 7F) is thicker than in the perinucleolar step; ist cells become cuboidal and attain a mean height of 5 μm. Acidophilic fibrils are located between the follicle cells in various positions (Figs. 4A,B and 7F), they are seen in circular, longitudinal, or spiral arrangements. The vascularized theca (Figs. 4B and 7F) is similar to that seen in the previous step. At the end of this step, the germinal vesicle has irregular folds (Figs. 3F and 7D,E).
Left illustration: Fig. 1. A: Crenichthys baileyi, adult female, USNM 391727; B: Empetrichthys latos, adult female, USNM 391728. Bar = 1 cm. Right illustration: Fig. 2. Ovary of Crenichthys baileyi. A and B: Panoramic views of the single ovary containing abundant follicles in different stages of development; the progressive enlargement in diameter of growing follicles is evident. The presence or absence of yolk and its amount differentiates the stages of oogenesis. Primary growth stage (PG), secondary growth stage (SG). A: H-E. Bar = 500 μm. B: H-E. Bar = 200 μm. C: The germinal epithelium (GE) borders the ovarian lumen (L). There are oogonia (OG) that are clearly differentiated from somatic epithelial cells because of their round shape, light ooplasm, and spherical nucleus with one nucleolus. Oocytes during primary growth (PG) are seen. Blood vessels (v) are seen in the stroma (S) that is subjacent to the germinal epithelium. PAS/MY-H. Bar = 10 μm. D and E: Germinal epithelium with an oogonium, (OG). Oocytes during chromatin nucleolus stage (CN), defined by the nucleus with chromosomes as fine filaments during early prophase I of meiosis. Oocytes in primary growth with basophilic ooplasm during the one nucleolus step (PGon), and multiple nucleoli step (PGmn) with a clear increase in diameter. Balbiani bodies (bb) are seen. Follicle cells (F) surround the oocytes. D: H-E. Bar = 12 μm, E: PAS/MY-H. Bar = 12 μm. F: Oocytes in multiple nucleoli step of primary growth (PGmn), presenting progressive increase in the number of nucleoli. Balbiani bodies (bb) are seen around the germinal vesicle. H-E. Bar = 50 μm.
Left illustration: Fig. 3. Oocytes during the primary growth stage in ovary of Crenichthys baileyi. A: Oocytes in the perinucleolar step (PGpn) of primary growth. The germinal vesicle is spherical and large. Most of the large nucleoli (nu) become oriented around the inner membrane of the germinal vesicle (gv). Balbiani bodies (bb) are seen. H-E. Bar = 50μm. Oocytes during the primary growth stage in ovary of Crenichthys baileyi. A: Oocytes in the perinucleolar step (PGpn) of primary growth. The germinal vesicle is spherical and large. Most of the large nucleoli (nu) become oriented around the inner membrane of the germinal vesicle (gv). Balbiani bodies (bb) are seen. H-E. Bar = 50 μm. B: Oocytes during the oil droplets step (PGod) of primary growth. The germinal vesicle (gv) contains numerous nucleoli (nu). H-E. Bar = 50 μm. C: Oocyte during the oil droplets step (PGod). The germinal vesicle (gv) is situated at the center of the oocyte. The ooplasm contains scarce oil droplets (od). H-E. Bar = 50 μm. D: Oocyte during the oil droplets step (PGod). The germinal vesicle (gv) presents numerous nucleoli (nu). There is a progressive increase in the number of oil droplets (od) and cortical alveoli (ca). Peripheral acidophilic fibrils (fb) are distributed between the follicle cells. H-E. Bar = 50 μm. E: Oocyte during the oil droplets step (PGod). Oil droplets (od) and cortical alveoli (ca) are more numerous. PAS/MY-H. Bar = 50 μm. F: At the end of oil droplets step (PGod), the germinal vesicle (gv) has irregular folds. The oil droplets (od) are located throughout the ooplasm. H-E. Bar = 100 μm. Right illustration: Fig. 4. Zona pellucida, follicle cells and theca of the follicle complex of Crenichthys baileyi. A–D: There is an increase in the thickness of the zona pellucida (zp) between the oil droplets step (PGod) of primary growth and the early (SGe) and late steps (SGl) of secondary growth. Oil droplets (od) and early deposition of yolk globules (y) is seen. Columnar follicle cells (F) with acidophilic fibrils (fb) are distributed between them. The theca (T) contains blood vessels (v). A: H-E. Bar = 25 μm. B: H-E. Bar = 25 μm. C: PAS/MY-H. Bar = 10 μm. D: H-E. Bar = 25 μm. B: Oocytes during the oil droplets step (PGod) of primary growth. The germinal vesicle (gv) contains numerous nucleoli (nu). H-E. Bar = 50 μm. C: Oocyte during the oil droplets step (PGod). The germinal vesicle (gv) is situated at the center of the oocyte. The ooplasm contains scarce oil droplets (od). H-E. Bar = 50 μm. D: Oocyte during the oil droplets step (PGod). The germinal vesicle (gv) presents numerous nucleoli (nu). There is a progressive increase in the number of oil droplets (od) and cortical alveoli (ca). Peripheral acidophilic fibrils (fb) are distributed between the follicle cells. H-E. Bar = 50 μm. E: Oocyte during the oil droplets step (PGod). Oil droplets (od) and cortical alveoli (ca) are more numerous. PAS/MY-H. Bar = 50 μm. F: At the end of oil droplets step (PGod), the germinal vesicle (gv) has irregular folds. The oil droplets (od) are located throughout the ooplasm. H-E. Bar = 100 μm. Right: Fig. 4. Zona pellucida, follicle cells and theca of the follicle complex of Crenichthys baileyi. A–D: There is an increase in the thickness of the zona pellucida (zp) between the oil droplets step (PGod) of primary growth and the early (SGe) and late steps (SGl) of secondary growth. Oil droplets (od) and early deposition of yolk globules (y) is seen. Columnar follicle cells (F) with acidophilic fibrils (fb) are distributed between them. The theca (T) contains blood vessels (v). A: H-E. Bar = 25 μm. B: H-E. Bar = 25 μm. C: PAS/MY-H. Bar = 10 μm. D: H-E. Bar = 25 μm.
Secondary Growth Stage or Vitellogenesis
The SG stage comprises: early secondary growth step (SGe), late secondary growth step (SGl), and full-grown oocyte step (SGfg). Early secondary growth step (SGe). The oocytes grow to a great extent through the active deposition of yolk, attaining a diameter of 1,200 μm. Folding of the germinal vesicle continuous at ist periphery (Fig. 8A) that began in the previous step. The oil droplets are abundant and located throughout the ooplasm (Figs. 5A and 8A). Early spherical, deeply acidophilic yolk vesicles, with a diameter of 2–5 μm, begin to accumulate at the oocyte periphery (Figs. 4C and 5A). The yolk vesicles progressively increase in number and size. Throughout this step, the yolk vesicles begin to fuse to form ovoid or irregular yolk globules (Fig. 8A). The zona pellucida is 15–16 μm thick and is a well-differentiated, striated layer between the oocyte and the follicle cells (Fig. 4B,C). The follicle cells remain as a single layer throughout SG, but become columnar, attaining 12–14 μm in height (Fig. 4C). The acidophilic fibrils (Fig. 4B–D), located between the follicular cells, progressively increase in number. The vascularized theca (Fig. 4C) is similar to that described earlier. Late secondary growth step (SGl). The germinal vesicle is eccentric and deeply folded along its periphery (Figs. 5B,C and 8B). The fusion of the yolk globules advances to form large regions of fluid yolk within the ooplasm (Figs. 5B–D and 8B– F). The zona pellucida attains 20 μm and appears clearly striated (Figs. 4D, 5C, and 8D–F). The micropyle was observed in the zona pellucida of oocytes of E. latos (Fig. 9A,B). The micropyle is a well-defined, channel-shaped opening. At its exterior edge, close to the follicle cell layer, the micropyle is 4 μm in diameter. It is slightly smaller at the interior edge of the zona pellucida, near the ooplasm, where its diameter is 3 μm (Fig. 9B). The sides of the micropyle are undulated, following the striated structure of the zona pellucida. The layer of follicle cells remains single, but decreases slightly in height, to 10–12 μm, compared with that observed in the previous step. The acidophilic fibrils are in the same position between the follicle cells (Figs. 5C and 8D–F). The theca (Figs. 5C and 8D) is similar to that described previously. Full-grown oocyte step (SGfg). The oocytes reach their maximum diameter, attaining 1.8–2 mm, in both species. The yolk globules are fused completely and form a large fluid globule of yolk (Figs. 5E,F and 9C). Oil droplets and cortical alveoli encircle the periphery of the ooplasm (Figs. 5E,F and 9C,D). The zona pellucida is 18–20 μm thick, and there are no evident striations (Fig. 9D). The follicular cells form a layer of 8–10 μm, slightly smaller than those seen in the previous step. The acidophilic fibrils are seen (Fig. 9D). The theca is similar to that described previously. Some full-grown oocytes were shrunken and the zona pellucida, follicle cells, and theca were separated from the oocyte by a space (Fig. 5E). Such shrinkage was likely due to formalin or Bouin’s fixation or the embedding medium. Throughout oogenesis, the germinal vesicle undergoes discrete morphological changes (Fig. 10A–D). Initially, during OP and CN stages, and PGon, PGmn (Fig. 10A), and PGpn steps, the germinal vesicle is spherical or ovoid in shape. Then, during the PGod step (Fig. 10B), it becomes slightly irregular in shape. Small and large nucleoli are seen. Finally, in the SG stage (Fig. 10C,D), it has an elongate, irregular shape, and its envelope becomes deeply folded. Small and large nucleoli are also seen. Postovulatory follicle complexes (Fig. 5A) in both species indicate that the specimens were collected during the spawning season.
Left illustration: Fig. 5. Oocytes during the secondary growth stage in ovary of Crenichthys baileyi. A: Oocyte during early secondary growth step (SGe) when the early deposition of yolk globules (y) appears in the ooplasm. The yolk globules are among abundant oil droplets (od). An oocyte during late secondary growth step (SGl) and a postovulatory follicle complex (POC) are also seen. H-E. Bar = 100 μm. Oocytes during the secondary growth stage in ovary of Crenichthys baileyi. A: Oocyte during early secondary growth step (SGe) when the early deposition of yolk globules (y) appears in the ooplasm. The yolk globules are among abundant oil droplets (od). An oocyte during late secondary growth step (SGl) and a postovulatory follicle complex (POC) are also seen. H-E. Bar = 100 μm. B and C: Two magnifications of an oocyte during late secondary growth step (SGl) when yolk globules (y) progressively fuse. The germinal vesicle (gv) is eccentric, and its periphery has irregular folds. The zona pellucida (zp), follicle cells with fibrils (fb) between them, and theca (T) surround the follicle. B: H-E. Bar = 100 μm. C: H-E. Bar = 50 μm. D: Oocyte during late secondary growth step (SGl) presenting clear increase of the yolk (y) fusion. H-E. Bar = 250 lm. E and F: The oocyte has reached ist maximum size during the full-grown oocyte step (SGfg). The smaller yolk globules become one large yolk globule (ygl) that occupies most of the oocyte volume. Oil droplets (od) and cortical alveoli (ca) are at the periphery of the ooplasm. The zona pellucida (zp) and theca (T) surround the follicle. E: PAS/MY-H. Bar = 250 μm. F: PAS/MY-H. Bar = 50 μm. Right illustration: Fig. 6. Ovary of Empetrichthys latos. A and B: Panoramic views of a single ovary containing abundant follicles in different stages of development, the progressive enlargement in diameter of the follicles is evident as they grow. The presence or absence of yolk and its amount differentiates the stages of oogenesis. Primary growth stage (PG), secondary growth stage (SG). A: PAS/MY-H. Bar = 500 μm. B: H-E. Bar = 200 μm. C: The germinal epithelium (GE) borders the ovarian lumen (L). Within it, oogonia (OG) possess spherical nuclei and one nucleolus. An oocyte during the perinucleolar step (PGpn) of primary growth is also seen, the germinal vesicle (gv) is large and spherical, with the nucleoli (nu) oriented peripherally. Squamous follicle cells (F) are seen. The stroma (S) is subjacent to the germinal epithelium. H-E. Bar = 10 μm. D: Oocytes in primary growth during the steps of one nucleolus (PGon), and multiple nucleoli (PGmn). The increase of ooplasmic basophilia is evident in multiple nucleoli (PGmn) by intense stained with hematoxylin. Balbiani bodies (bb) are seen near the germinal vesicle. H-E. Bar = 10 μm.
DISCUSSION
The histological analysis of the ovary and oogenesis in C. baileyi and E. latos, species so rare in the wild, permitted this description and documentation of essential aspects of their reproduction. Here, we summarize and compare the earlier suite of reproductive characters among the two subfamilies of goodeid and the oviparous Fundulus, first describing the characters that all three atherinomorph taxa share (Parenti, 2005), and then those characters that vary and allow us to make statements about relationship. Character states that vary are also tabulated (Table 1).
Shared Characters
Germinal epithelium. Activity of the germinal epithelium underlies the seasonality of the female reproductive cycle. The germinal epithelium lines the ovarian lumen and is the location of the oogonia, which divide by mitosis, proliferate and develop into the next generation of germ cells during the reproductive cycle (Grier, 2000). Oogonia divide mitotically and become oocytes when they enter meiosis. Mitosis of oogonia is the first stage (OP) in the classification of oogenesis in teleosts presented by Grier et al. (2009), and is documented here in the oogenesis of C. baileyi and E. latos. A germinal epithelium has also been documented in Fundulus and several species of viviparous goodeids (Parenti & Grier, 2004; Grier et al., 2005). Follicle cells. Follicle cells remain as a single layer throughout oogenesis. Changes in follicle cells during follicle development in C. baileyi and E. latos range from being a squamous layer during PG to cuboidal or columnar cells during SG, with a reduction to cuboidal or squamous again in fullgrown follicles. These changes are similar to those described in viviparous goodeids (Mendoza, 1965; Uribe et al., 2004, 2005, 2009). Likewise, similar morphological changes were described in the oviparous cyprinodontiforms Fundulus heteroclitus by Matthews (1938) and Cynolebias melanotaenia and Cynolebias ladigesi by Wourms (1976). The increase in follicle cell size is related to the active process of vitellogenesis during SG. The follicle cells are involved in the synthesis of a diversity of proteins and lipids during the growth of the oocytes and, consequently, they become smaller once vitellogenesis is complete (Guraya, 1986). Folliculogenesis. Folliculogenesis in C. baileyi and E. latos is complete when a layer of prefollicle cells, derived from the somatic epithelial cells of the germinal epithelium, entirely enclose each oocyte to form follicle cells that are surrounded by a basement membrane (Grier, 2000; Grier et al., 2005, 2009). The follicle consists of an oocyte and surrounding follicle cells (Grier, 2000). The follicle is enclosed by a basement membrane and theca to form the ovarian follicle complex. Development of the follicle is included in the second stage (CN) of the classification of oogenesis (Grier et al., 2009), and continues into PG. Descriptions of the elements of folliculogenesis are similar in many teleosts (for reviews see, Dodd & Sumpter, 1984; Selman and Wallace, 1989). The stages and steps developed during oogenesis in C. baileyi and E. latos are similar to those reported for other atherinomorphs, including Fundulus and viviparous goodeids. Cystovarian condition. The ovary of C. baileyi and E. latos is cystovarian; it has a central lumen of coelomic origin. The ovarian wall possesses ovigerous folds or lamellae that are lined by germinal epithelium and project into the lumen (Dodd & Sumpter, 1984; Grier, 2000). The lamellae contain ovarian follicles in different stages of development and the mature oocytes are ovulated into the ovarian cavity, features documented in several other cyprinodontiform taxa, including Fundulus (Matthews, 1938; Brummett et al., 1982; Dodd & Sumpter, 1984; Guraya, 1986; Grier, 2000; Parenti & Grier, 2004) and viviparous goodeids (Grier et al., 2005, 2009). Primary growth stage. The initial steps of PG, common not only to teleosts but also other vertebrates that develop yolked oocytes, are characterized by basophilia of the ooplasm. This indicates a period of intense RNA synthesis coupled with ribosome production to support development of the oocyte and the embryo (Wallace & Selman, 1990; Patiño & Sullivan, 2002; Grier et al., 2009). Initially, during PG, Balbiani bodies are seen around the periphery of the germinal vesicle; they subsequently migrate to the oocyte periphery and disperse. Balbiani bodies have been observed in previtellogenic oocytes in viviparous goodeids (Uribe et al., 2005, 2009) as well as other viviparous and oviparous species (Droller & Roth, 1966; Azevedo, 1984; Guraya, 1986; Kobayashi & Iwamatsu, 2000). During PG, cortical alveoli and oil droplets develop gradually in relation to the increase in oocyte diameter (Selman et al., 1988). This process is similar not just in atherinomorphs but numerous other teleosts (for reviews see, Dodd & Sumpter, 1984; Selman & Wallace, 1989; Tyler & Sumpter, 1996; Grier et al., 2009). The initial oil droplets are located around the germinal vesicle in both C. baileyi and E. latos, agreeing with the description in F. heteroclitus by Selman et al. (1988) and Selman and Wallace (1989), viviparous goodeids and numerous other teleost species (for reviews see Guraya, 1986; Grier et al., 2009). The similar morphology of cortical alveoli and oil droplets observed in C. baileyi and E. latos, during the early oil droplets step of primary growth (PGod) make their differentiation difficult. This observation agrees with those of Selman & Wallace (1989) and Tyler & Sumpter (1996), who both indicated that oil droplets are difficult to distinguish from cortical alveoli because the contents of both are leached out because of inadequate fixation in the histological process. In addition, the empetrichthyine specimens examined here were preserved in alcohol for nearly half a century, which may have compromised the integrity of the oil droplets and cortical alveoli. Secondary growth stage. Oocytes of C. baileyi and E. latos enlarge greatly to attain their maximum diameter during SG due to the synthesis and accumulation of yolk. Yolk is the fundamental material stored in the ooplasm during SG, as in other teleosts (Guraya, 1986; Wallace & Selman, 1990). Yolk is used for the nutrition and metabolic activities during embryonic development. Yolk proteins are derived from vitellogenin, the hepatic yolk precursor. Vitellogenin is a glycolipophosphoprotein transported by blood vessels from the liver to the ovarian follicle and is taken up by developing oocytes. Vitellogenin enters the oocytes by binding to specific receptors on the oolemma. Then, it is internalized by endocytosis. Once in theooplasm, vitellogenin is divided into smaller molecular weight polypeptides: the yolk proteins lipovitellin, phosvitin, and bβ´-component. The yolk proteins are then packed into yolk globules, which are stored in developing oocytes, and used as an energy source for embryonic development (Patiño & Sullivan, 2002; LaFleur et al., 2005; Raldúa et al., 2006; Grier et al., 2009). During SG, yolk fuses in C. baileyi and E. latos, similar to that described in the species of viviparous goodeids (Mendoza, 1943; Wourms, 1981; Schindler & Hamlett, 1993; Uribe et al., 2004, 2005, 2009). Yolk is fluid also in F. heteroclitus (see Matthews, 1938; Selman & Wallace, 1983; Selman et al., 1986) and in other atherinomorphs (Wallace & Selman, 1980, 1981; Guraya, 1986; Selman et al., 1986; Selman & Wallace, 1989; Parenti & Grier, 2004; Jalabert, 2005; Grier et al., 2009). This may result from a deep reorganization of the lipoprotein yolk involving the action of proteolytic enzymes, as suggested by Jalabert (2005). Fluid yolk is one diagnostic character of atherinomorphs that supports their monophyly (Parenti & Grier, 2004).
Left illustration: Fig. 7. Oocytes during the primary growth stage in ovary of Empetrichthys latos. A–D: Oocytes during oil droplets step (PGod) of primary growth clearly illustrate the increase of the number of oil droplets (od) and cortical alveoli (ca). The germinal vesicle (gv) is changing from spherical and centrally located to irregular in shape and eccentric. A: H-E. Bar = 50 μm. B: H-E. Bar = 50 μm. C: PAS/MY-H. Bar = 50 μm. D: PAS/MY-H. Bar = 100 μm. E and F: At the end of oil droplets step (PGod), the germinal vesicle (gv) is eccentric, and its periphery has irregular folds. The oil droplets (od) are located throughout the ooplasm. The zona pellucida (zp) is well defined. Peripheral acidophilic fibrils (fb) are distributed between the follicle cells (F). The theca (T) contains blood vessels (v). E: H-E. Bar = 50 μm. F: H-E. Bar = 25 μm. Right illustration: Fig. 8. Oocytes during the secondary growth stage in ovary of Empetrichthys latos. A: Oocytes during early secondary growth step (SGe) when the early deposition of yolk globules (y) appears in the ooplasm. The yolk globules are located among abundant oil droplets (od). The germinal vesicle (gv) is eccentric, and its periphery has irregular folds. PAS/MY-H. Bar = 100 μm. B–F: Oocytes during the late secondary growth step (SGl) when yolk globules (y) progressively fuse. Oil droplets and cortical alveoli (ca) are seen. The germinal vesicle (gv) remains eccentric. The yolk globules become one large yolk globule (ygl) that occupies most of the oocyte volume. The zona pellucida (zp), follicle cells (F), and theca (T) surround the follicle. The zona pellucida (zp) thickens between the oil droplets step (PGod) of primary growth, early secondary growth (SGe), and late secondary growth steps (SGl). Fibrils (fb) distributed between the follicle cells are observed. B: PAS/MY-H. Bar = 100 μm. C: PAS/MY-H. Bar = 50 μm. D: PAS/ MY-H. Bar = 10 μm. E: PAS/MY-H. Bar = 20 μm. H-E. F: Bar = 10 μm.
Left illustration: Fig. 9. Oocytes during the secondary growth stage in ovary of Empetrichthys latos. A and B: Micropyle in an oocyte in late secondary growth step (SGl). The micropyle (mi) opening in the zona pellucida (zp) is slightly smaller at the interior edge near the ooplasm (oop). Follicle cells (F) surround the zona pellucida. A: H-E. Bar = 10 μm. B: H-E. Bar = 12 μm. C and D: The oocyte reaches its maximum size during the full-grown oocyte step (SGfg). The yolk globules fuse and become one large yolk globule that occupies most of the oocyte volume. Oil droplets (od) and cortical alveoli (ca) are at the periphery of the ooplasm. The zona pellucida (zp) increases thickness as oogenesis advances, and the striation is not seen. Acidophilic fibrils (fb) distributed between the follicle cells (F) are observed. C: H-E. Bar = 100 μm. D: H-E. Bar = 12 μm. Right illustration: Fig. 10. Sequence of morphological changes of the germinal vesicle during oogenesis in Crenichthys baileyi (A,C) and Empetrichthys latos (B,D). A and B: In oocytes in primary growth stage, during multiple nucleoli step (PGmn) and oil droplets step (PGod), having a spherical shape. Large nucleoli (Nu) and small nucleoli (nu) are seen in the germinal vesicle. A: H-E. Bar = 10 μm. B: H-E. Bar = 10 μm. C and D: In oocytes in secondary growth stage, during early secondary growth step (SGe) and late secondary growth step (SGl), having the germinal vesicle deeply folded and large peripheral nucleoli. There are also numerous small nucleoli (nu) that are less peripheral than the large ones (Nu). Lampbrush chromosomes (ch) are also seen. C: H-E. Bar = 10 μm. D: H-E. Bar = 10 μm.
Characters that vary
Ovarian structure. Most teleosts have paired ovaries suspended via a mesovarium from the dorsal wall of the coelom. A single, median ovary is typical of viviparous and some oviparous teleosts (Grier et al., 2009). All viviparous goodeids and C. baileyi and E. latos have a single, median ovoid ovary with no obvious external traces of fusion. A single ovary, resulting from fusion of both ovaries during embryological development, has been documented in viviparous goodeids by numerous authors, including Hubbs & Turner (1939), Turner, (1947), Amoroso (1960), Mendoza (1965), Wourms (1981), Dodd & Sumpter (1984), Wourms et al. (1988), and Schindler & Hamlett (1993). The ovary of F. heteroclitus was described by Matthews (1938, p 70) as unpaired, however, bilobed anteriorly with a mid-ventral groove that separates it superficially into right and left portions. Ovarian septum. The ovary has an internal septum that divides the ovarian lumen into two compartments in all species of viviparous goodeids (Turner, 1947; Mendoza, 1965; Wourms, 1981; Schindler & Hamlett, 1993; Uribe et al., 2004, 2005, 2009). Gestation occurs in both chambers of the ovarian lumen (Uribe et al., 2005). There is no ovarian septum in the oviparous C. baileyi and E. latos, or in F. heteroclitus (see Matthews, 1938). Oocyte size. Egg size is a direct indicator of fish development (Elinson, 1989). There is a distinct difference in egg size between species of the two subfamilies of goodeids. The maximum oocyte diameter in both C. baileyi and E. latos is 1.8–2 mm. The oocyte diameter of the oviparous F. heteroclitus is 1.7–1.9 mm (Selman & Wallace, 1983; Selman et al., 1986), similar to that observed here, and in Kopec (1949), for C. baileyi and E. latos. In contrast, the maximum diameter of oocytes of viviparous goodeids is much smaller: 1.0 mm in Ataeniobius toweri (Turner, 1940), 0.8 mm in Goodea atripinnis, Characodon lateralis (Turner, 1940; Uribe et al., 2005), and "Xenotoca" eiseni (Mendoza, 1965), 0.7 mm in Ilyodon whitei (Uribe et al., 2004, 2005), and 0.5 mm in Neotoca bilineata (Turner, 1933), for example. The reduced diameter of full-grown oocytes in viviparous goodeids is related to the intraluminal gestation in which embryos obtain nutrients from the mother, a process known as matrotrophy. These additional nutrients are contained in the histotrophe secreted by the ovarian epithelium into the ovarian lumen (Turner, 1937, 1947; Amoroso, 1960; Mendoza, 1972; Wourms, 1981, 2005; Schindler & De Vries, 1987). During gestation, transfer of nutrients from the mother to the embryo occurs through different means. The histotrophe may be absorbed by the trophotaeniae, extensions of the hindgut of the embryo into the ovarian lumen that are lined with absorptive epithelium (Turner, 1937; Wourms, 1981, 2005; Wourms et al., 1988). Schindler & Greven (1992) documented endocytosis of proteins from the histotrophe by the trophotenial absortive cells in the viviparous goodeid Ameca splendens. Also, embryos may be oophagous (they eat and digest eggs from the ovarian lumen) or adelphophagous (they eat and digest other embryos; Greven & Großherr, 1992). Cortical alveoli. Cortical alveoli are observed in Goodeinae and Empetrichthyinae species, but in the latter, cortical alveoli are larger, sometimes attaining 50 μm in diameter. Large cortical alveoli have also been documented in F. heteroclitus (see Anderson, 1968; Selman et al., 1988; Wallace & Selman, 1990). Fluid yolk. Yolk is fluid throughout the oocyte during SG in C. baileyi and E. latos as described also in viviparous goodeids and in all other atherinomorph fishes as far as known (Parenti & Grier, 2004; Parenti, 2005). In viviparous goodeids, fluid yolk comprises most of the ooplasm (Wourms et al., 1981; Uribe et al., 2005). In contrast, in C. baileyi and E. latos, as in F. heteroclitus, the oocyte also has oil droplets at its periphery (Matthews, 1938; Wallace & Selman, 1990). Zona pellucida. The zona pellucida in fullgrown oocytes of C. baileyi and E. latos is 18–20 μm thick, comparable with the thickness recorded in F. heteroclitus by Brummett (1966) and Kuchnow & Scott (1977). The increase in the number of microvilli, which gives the zona pellucida ist striated appearance during oocyte growth, amplifies the surface of interchange between oocyte and follicle cells. During oogenesis, the germ cells and follicle cells remain connected by a variety of adhering junctional complexes. Intercellular communication by gap junctions between the oocyte and follicle cells during OM in F. heteroclitus was suggested by Cerda` et al. (1993) to have an important functional role in the transfer of various substances. They further propose that such communication is necessary for the maintenance of meiotic arrest and hydration that occurs by the translocation from follicle cells to maturing oocyte of K1, the primary osmotic effector for oocyte hydration. During OM, oocyte microvilli may decrease in number mitigating the striated appearance of the zona pellucida (Cerdà et al., 1999). Similar changes in the zona pellucida surrounding oocytes in late oogenesis were reported in the oviparous seahorse, Hippocampus erectus and pipefish, Syngnathus fuscus by Anderson (1967), and croaker, Micropogonias undulatus by York et al. (1993), and also in a viviparous halfbeak, Dermogenys pusillus by Flegler (1977). Reduction of the striated appearance of the zona pellucida in F. heteroclitus was described by Kuchnow & Scott (1977). In addition, Dumont & Brummett (1980) suggested that the changes in the structure of the zona pellucida of F. heteroclitus may be related to changes in the oocyte volume through hydration during maturation. In contrast to the thick zona pellucida of oviparous species, a thin zona pellucida is formed by the follicle of viviparous species (Flegler, 1977; Uribe et al., 2005, 2009). The zona pellucida of oocytes, during late SG step, in viviparous goodeids, such as I. whitei and G. atripinnis, reaches 2–3 μm in thickness (Uribe et al., 2009). Micropyle. A single micropyle, an opening in the zona pellucida at the animal pole through which the sperm enters the oocyte, is a synapomorphy of the Actinopterygii, the ray-finned fishes (Bartsch & Britz, 1997). A micropyle in the zona pellucida of F. heteroclitus was documented by Kuchnow & Scott (1977) and Dumont & Brummett (1980), and in Fundulus grandis by Grier et al. (2009). A micropyle was observed in E. latos (Fig. 9A,B) and is surrounded by a striated zona pellucida indicating that it forms before OM, during late SG. A micropyle was not seen in C. baileyi. Although widely reported in oviparous species, a micropyle has never been documented in a viviparous species (Grier et al., 2009; Uribe et al., 2009). In viviparous goodeids, the sperm enters the oocyte through a ‘‘delle’’ (named by Stuhlmann, 1887): a thin, funnel-like channel that connects the ovarian lumen with the oocyte. A micropyle is a structure in the zona pellucida through which a sperm pass to reach the oocyte surface. A delle is a passageway from the ovarian lumen to the ovum formed by cells of the germinal epithelium. Sperm reach the oocyte through a small opening in the epithelium at the base of the delle (see Bailey, 1933, pp 207–208) and presumably pass through a micropyle to reach the oocyte surface; fertilization is intrafollicular. The presence of a micropyle has yet to be demonstrated in viviparous goodeids. Homology of a micropyle and an opening at the base of the delle is not endorsed here and is a topic for further study. Fibrils. Elongate, adhesive fibrils or filaments between the follicular cells on the surface of developing oocytes characterize both C. baileyi and E. latos. Fibrils are formed by the follicle cells (Anderson, 1967; Dumont & Brummett, 1980). Fibrils or filaments over all or a portion of the egg surface are characteristic of many other oviparous cyprinodontiform species such as F. heteroclitus, has been documented extensively (e.g., Kemp & Allen, 1956; Shanklin, 1959; Brummett, 1966; Kuchnow & Scott, 1977; Dumont & Brummett, 1980; Brummett & Dumont, 1981). Fertilized eggs adhere to vegetation via fibrils. Fibrils may also protect the egg as they trap debris and obscure the embryo from view (Brummett, 1966). They may also help to prevent water loss during low tide when the embryo may be exposed (Dumont & Brummett, 1980). Eggs of all viviparous species lack fibrils on the surface of the zona pellucida (Uribe et al., 2005, 2009; Parenti et al., 2010). Absence of fibrils is interpreted as an evolutionary loss of a structure that is no longer needed in fishes in which development is intraovarian.
CONCLUSIONS
Goodeid embryos develop in two different environments: the exterior in the oviparous species and within the ovarian lumen in viviparous species. Size and external surface features of the egg reflect the environment in which the embryo develops (Guraya, 1986). Eggs of the oviparous Crenichthys and Empetrichthys are relatively large and covered with adhesive fibrils or filaments. They have a thick zona pellucida. A micropyle was seen only in E. latos. They share these characters with other oviparous taxa, such as species in the genus Fundulus discussed here. Viviparous goodeids have relatively small eggs, no adhesive fibrils, and a delle rather than a micropyle, characters consistent with their reproductive mode. Also, cortical alveoli are large in Empetrichthys, Crenichthys, and Fundulus relative to those in viviparous goodeids. A single derived reproductive character is shared by all viviparous goodeids and C. baileyi and E. latos: a single, median ovoid ovary with no obvious external evidence of fusion. This is in contrast to the oviparous Fundulus which has a single ovary separated only superficially by a mid-ventral groove into right and left portions (Matthew, 1938). A single, median ovary characterizes all viviparous and some oviparous cyprinodontiforms, as well as other teleosts (Grier et al., 2009). We interpret this character as a synapomorphy of the Goodeidae; however, because the character is so widespread among teleosts, we hesitate to infer that it represents an intermediate state between oviparity and viviparity. Other reproductive characters, especially those of males, need to be surveyed before we can hypothesize about the stages in evolution of viviparity among goodeids.